Abstract
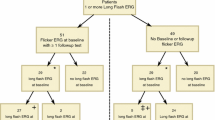
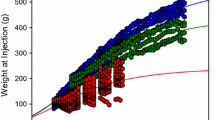
Electroretinograms (ERGs) have been recorded longitudinally in children before and during treatment with the antiepileptic drug vigabatrin for the past 3.5 years. Vigabatrin induced changes in ERG responses occur in children; the most dramatic changes occur in the oscillatory potentials. The purpose of this study was to identify changes in ERG responses associated with discontinuation of vigabatrin treatment. If vigabatrin-induced changes reverse after discontinuation of the drug we infer that the original change is not an indicator of toxicity. ERG data were analyzed from 17 children who discontinued vigabatrin therapy. The duration of treatment ranged from 5 to 52 months, the age for the first ERG ranged from 6 to 38 months (median 10 months). ERGs were tested using the standard protocol established by the International Society for Clinical Electrophysiology of Vision, with Burian-Allen bipolar contact-lens electrodes. In addition to standard responses we recorded photopic oscillatory potentials (OPs). During vigabatrin treatment OPs show a greater change than other ERG responses, with the early occurring wavelets from the photopic OPs showing the greatest change. With discontinuation of vigabatrin the amplitude of the early wavelets of the photopic OPs increased dramatically compared with amplitudes while taking the drug (paired t-test, p=0.000075). The scotopic oscillatory potentials also show some recovery. Although changes in oscillatory potentials may occur with vigabatrin toxicity, a large change likely occurs with a non-toxic pharmacological effect of vigabatrin on GABAergic amacrine cells in the inner plexiform layer. Reduction of OPs in children on vigabatrin may not be related to toxicity.
Similar content being viewed by others
References
Fois A, Buoni S, Bartolo R, Marco V, Mostardini R. Vigabatrin treatment in children. Child's Nerv Syst 1994; 10: 244–8.
Kwong L. Vigabatrin as first line therapy in infantile spasms: review of seven patients. J Paediatr Child Health 1997; 33(2): 121–4.
Uldall P, Alving J, Gram L, Beck S. Vigabatrin in pediatric epilepsy - An open study. J Child Neurol 1991; 6: 2S38–44.
Uldall P, Alving J, Gram L, Hogenhaven H. Vigabatrin in Childhood Epilepsy: A 5-Year Follow-Up Study. Neuropediatrics 1995; 26: 253–6.
Nabbout R, Melki I, Gerbaka B, Dulac O, Akatcherian C. Infantile spasms in Down syndrome: good response to a short course of vigabatrin. Epilepsia 2001; 42(12): 1580–3.
Mackay M, Weiss S, Snead OC 3rd. Treatment of infantile spasms: an evidence-based approach. Int Rev Neurobiol 2002; 49: 157–84.
Elterman RD, Shields WD, Mansfield KA, Nakagawa J. Randomized trial of vigabatrin in patients with infantile spasms. Neurology 2001; 57(8): 1416–21.
Eke T, Talbot JF, Lawden MC. Severe persistent visual field constriction associated with vigabatrin. Br Med J 1997; 314: 180–1.
Lawden MC, Eke T, Degg C, Harding GF, Wild JM. Visual field defects associated with vigabatrin therapy [see comments]. J Neurol Neurosurg Psychiatry 1999; 67(6): 716–22.
Wild JM, Martinez C, Reinshagen G, Harding GF. Characteristics of a unique visual field defect attributed to vigabatrin. Epilepsia 1999; 40(12): 1784–94.
Kalviainen R, Nousiainen I, Mantyjarvi M, Nikoskelainen E, Partanen J, Partanen K, Riekkinen P Sr. Vigabatrin, a gabaergic antiepileptic drug, causes concentric visual field defects. Neurology 1999; 53(5): 922–6.
Manuchehri K. Visual field defect associated with vigabatrin. Methodof estimating prevalence was inappropriate [letter]. Br Med J 2000; 320(7246): 1403–4; discussion 1404.
van der Torren K, Graniewski-Wijnands HS, Polak BC. Visual field and electrophysiological abnormalities due to vigabatrin. Doc Ophthalmol 2002; 104(2): 181–8.
Hardus P, Verduin WM, Berendschot TT, Kamermans M, Postma G, Stilma JS, van Veelen CW. The value of electrophysiology results in patients with epilepsy and vigabatrin associated visual field loss. Acta Ophthalmol Scand2001; 79(2): 169–74.
Kalviainen R, Nousiainen I. Visual fieldd efects with vigabatrin: epidemiology and therapeutic implications. CNS Drugs 2001; 15(3): 217–30.
Russell-Eggitt IM, Mackey DA, Taylor DS, Timms C, Walker JW. Vigabatrin-associated visual field defects in children. Eye 2000; 14 (Pt 3a): 334–9.
Iannetti P, Spalice A, Perla FM, Conicella E, Raucci U, Bizzarri B. Visual field constriction in children with epilepsy on vigabatrin treatment. Pediatrics 2000; 106(4): 838–42.
Gross-Tsur V, Banin E, Shahar E, Shalev RS, Lahat E. Visual impairment in children with epilepsy treated with vigabatrin. Ann Neurol 2000; 48(1): 60–4.
Luchetti A, Amadi A, Gobbi G. Visual field defects associated with vigabratin monotherapy in children. J Neurol Neurosurg Psychiatry 2000; 69(4): 566.
Daneshvar H, Racette L, CouplandSG, Kertes PJ, Guberman A, Zackon D. Symptomatic and asymptomatic visual loss in patients taking vigabatrin. Ophthalmology 1999; 106(9): 1792–8.
Krauss GL, Johnson MA, Miller NR. Vigabatrin-associated retinal cone system dysfunction: electroretinogram and ophthalmologic findings [see comments]. Neurology 1998; 50(3): 614–8.
Miller NR, Johnson MA, Paul SR, Girkin CA, Perry JD, Endres M, Krauss GL. Visual dysfunction in patients receiving vigabatrin: clinical and electrophysiologic findings. Neurology 1999; 53(9): 2082–7.
Arndt CF, Derambure P, Defoort-Dhellemmes S, Hache JC. Outer retinal dysfunction in patients treated with vigabatrin. Neurology 1999; 52(6): 1201–5.
Harding GF, Wild JM, Robertson KA, Rietbrock S, Martinez C. Separating the retinal electrophysiologic effects of vigabatrin: treatment versus field loss [In Process Citation]. Neurology 2000; 55(3): 347–52.
Besch D, Kurtenbach A, Apfelstedt-Sylla E, Sadowski B, Dennig D, Asenbauer C, Zrenner E, Schiefer U. Visual field constriction and electrophysiological changes associated with vigabatrin. Doc Ophthalmol 2002; 104 (2): 151–70.
Jensen H, Sjo O, Uldall P, Gram L. Vigabatrin and retinal changes. Doc Ophthalmol 2002; 104(2): 171–80.
Westall CA, Logan WJ, Smith K, Buncic JR, Panton CM, Abdolell M. The Hospital for Sick Children, Toronto, Longitudinal ERG study of children on vigabatrin. Doc Ophthalmol 2002; 104(2): 133–49.
Morong S, Nobile R, Westall CA, Buncic JR, Logan WJ, Panton CM, Abdolell M. Longitudinal changes in photopic OPs occurring with vigabatrin treatment. Doc Ophthalmol 2003; 107: 289–297.
Comaish IF, Gorman C, Brimlow GM, Barber C, Orr GM, Galloway NR. The effects of vigabatrin on electrophysiology and visual fields in epileptics: a controlled study with a discussion of possible mechanisms. Doc Ophthalmol 2002; 104(2): 195–212.
Harding GFA. Electrophysiological methods for assessing fieldlosses associated with Vigabatrin. in International Societry for Clinical Electrophysiology of Vision XXXVIII Symposium 2000. Sydney, Australia.
Johnson MA, Krauss GL, Miller NR, Medura M, Paul SR. Visual function loss from vigabatrin: effect of stopping the drug. Neurology 2000; 55(1): 40–5.
Graniewski-Wijnands HS, van der Torren K. Electroophthalmological recovery after withdrawal from vigabatrin. Doc Ophthalmol 2002; 104(2): 189–94.
Coupland SG, Zackon DH, Leonard BC, Ross TM. Vigabatrin effect on inner retinal function. Ophthalmology 2001; 108(8): 1493–6; discussion 1497-8.
Marmor MF, Zrenner E. Standard for clinical electroretinography (1999 update). Doc Ophthalmol 1999; 97: 143–56.
Westall CA, Panton CM, Levin AV. Time course for maturation of electroretinogram response, from infancy to adulthood. Doc Ophthalmol 1999; 96: 355–79.
Watchtmeister L. Oscillatory potentials in the retina: what do they reveal. Prog Retin Eye Res 1998; 17(4): 485–521.
Nelson R, Famiglietti EV Jr., Kolb H. Intracellular staining reveals different levels of stratification for on-and off-center ganglion cells in cat retina. J Neurophysiol 1978; 41(2): 472–83.
Grunert U. Distribution of GABA and glycine receptors on bipolar and ganglion cells in the mammalian retina. Microsc Res Tech 2000; 50(2): 130–40.
Kojima M, Zrenner E. Off-components in response to brief light flashes in the oscillatory potential of the human electroretinogram. Albrecht Von Graefes Arch Klin Exp Ophthalmol 1978; 206(2): 107–20.
Marc RE, Liu WL. (3H) glycine-accumulating neurons of the human retina. J Comp Neurol 1985; 232(2): 241–60.
Pourcho RG. Uptake of [3H]glycine and [3H]GABA by amacrine cells in the cat retina. Brain Res 1980; 198(2): 33–46.
Harding GF, Spencer EL, Wild JM, Conway M, Bohn RL. Field-specific visual-evoked potentials: identifying field defects in vigabatrin-treated children. Neurology 2002; 58(8): 1261–5.
Harding GF, Robertson KA, Edson AS, Barnes P, Wild J. Visual electrophysiological effect of a GABA transaminase blocker. Doc Ophthalmol 1999; 97(2): 179–88.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Westall, C.A., Nobile, R., Morong, S. et al. Changes in the electroretinogram resulting from discontinuation of vigabatrin in children. Doc Ophthalmol 107, 299–309 (2003). https://doi.org/10.1023/B:DOOP.0000005339.23258.8f
Issue Date:
DOI: https://doi.org/10.1023/B:DOOP.0000005339.23258.8f