Abstract
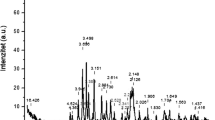
It has been found that crystalline BaCO3 replaces crystalline BaAl2O4 in prototypical NO x traps upon exposure to liquid water. This phenomenon can be understood in terms of acid–base chemistry, where Ba2+ is leached from the aluminate, precipitating as a carbonate, under the influence of weak carbonic acid naturally present due to the equilibrium between CO2 in the air and the water. Consequences of such a redistribution of Ba2+ in a NO x trap could include a decrease in surface area of the trapping material and a loss of contact between the trapping material and the catalytic components, such as Pt. Indeed, preliminary testing has shown that exposure of fully formulated NO x traps to liquid water results in the loss of approximately half of the initial trapping capacity.
Similar content being viewed by others
References
N. Takahashi, H. Shinjoh, T. Iijima, T. Suzuki, K. Yamazaki, K. Yokota, H. Suzuki, N. Miyoshi, S. Matsumoto, T. Tanizawa, T. Tanaka, S. Tateishi and K. Kasahara, Catal. Today 27 (1996) 63.
J.P. Breen, M. Marella, C. Pistarino and J.R.H. Ross, Catal. Lett. 80 (2002) 123.
J.R. Theis, J.A. Ura, J. Li, G.G. Surnilla, J.M. Roth and C.T. Goralski Society of Automotive Engineers Paper No. 2003-01-1159
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Graham, G., Jen, HW., Theis, J. et al. Leaching of Ba2+ in NOx Traps. Catalysis Letters 93, 3–6 (2004). https://doi.org/10.1023/B:CATL.0000016946.28770.37
Issue Date:
DOI: https://doi.org/10.1023/B:CATL.0000016946.28770.37