Abstract
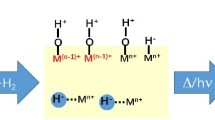
An alternative mechanism by which nitrous oxide is formed during both the catalytic reaction of nitric oxide and the oxidation of ammonia at platinum surfaces is suggested.
Similar content being viewed by others
References
R. Burch, Top. Catal. 24 (2003) 97.
D.W. Johnson, M.H. Matloob and M.W. Roberts, J. Chem. Soc., Faraday Trans. 1 75 (1979) 2143.
C.T. Au, A.F. Carley and M.W. Roberts, Philos. Trans. Roy. Soc. Lond. A 318 (1986) 61.
A.F. Carley and M.W. Roberts, Proc. Roy. Soc. Lond. A 363 (1978) 40.
T.S. Amorelli, A.F. Carley, M.K. Rajumon, M.W. Roberts and P.B. Wells Surf. Sci. 315 (1994) L900.
T. Greber, K. Freihube, R. Grobecker, A. Botcher, K. Hermann, G. Ertl and D. Fick Surf. Sci. 313 (1994) L806.
A.F. Carley, P.R. Davies and M.W. Roberts, Catal. Lett. 80 (2002) 25(and references therein).
D. Wang and M.A. Barteau, Catal. Lett. 90 (2003) 7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roberts, M. The Formation of N2O During the Chemisorptions of Nitric Oxide at Platinum Surfaces at Low Temperature: A Comment on the Mechanism. Catalysis Letters 93, 29–30 (2004). https://doi.org/10.1023/B:CATL.0000016942.07960.ba
Issue Date:
DOI: https://doi.org/10.1023/B:CATL.0000016942.07960.ba