Abstract
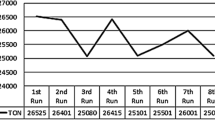
The H2 reduction of RuO2 hydrate “dissolved” in 1-n-butyl-3-methylimidazolium ionic liquids with different counterions, hexafluorophosphate (BMI ⋅ PF6), tetrafluoroborate (BMI ⋅ BF4) and trifluoromethane sulfonate (BMI ⋅ SO3CF3), is a simple and reproducible method for the preparation of ruthenium nanoparticles of 2.0–2.5 nm diameter size and with a narrow size distribution. The Ru nanoparticles were characterized by TEM and XRD. The isolated Ru nanoparticles are reoxidized in air, whereas they are less prone to oxidation when imbibed in the ionic liquids. These nanoparticles are active catalysts for the solventless or liquid–liquid biphasic hydrogenation of olefins under mild reaction conditions (4 atm, 75°C). The catalytic system composed of nanoparticles dispersed in BMI ⋅ PF6 ionic liquid is very stable and can be reused several times without any significant loss in the catalytic activity. Total turnover numbers greater than 110 000 (based on total Ru) or 320 000 (corrected for exposed Ru atoms) were attained within 80 h for the hydrogenation of 1-hexene.
Similar content being viewed by others
References
H. Over, Y.D. Kim, A.P. Seitsonen, S. Wendt, E. Lundgren, M. Schmid, P. Varga, A. Morgante and G. Ertl, Science 287 (2000) 1474; (b) L. Zang and H. Kisch, Angew. Chem., Int. Ed. 39 (2000) 3921; (c) A. Böttcher, M. Rogozia, H. Niehus, H. Over and G. Ertl, J. Phys. Chem., B 103 (1999) 6267.
A. Mills, Chem. Soc. Rev. 18 (1989) 285; (b) H. Harriman, Platinum Met. Rev. 27 (1983) 102; (c) W.D. An, J.K. Hong, P.N. Pintauro, K. Warner and W. Neff, J. Am. Oil Chem. Soc. 75 (1998) 917.
H. Madhavaram, H. Idriss, S. Wendt, Y.D. Kim, M. Knapp, H. Over, J. Aßmann, E. Löffler and M. Muhler, J. Catal. 202 (2001) 296.
K. Kaneda, S. Haruna, T. Imanaka and K. Kawamoto, J. Chem. Soc., Chem. Commun. (1990) 1467.
B.-Z. Zhan, M.A. White, T.-K. Sham, J.A. Pincock, R.J. Doucet, K.V.R. Rao, K.N. Robertson and T.S. Cameron, J. Am. Chem. Soc. (2003) in press.
See for example: R. Kikuchi, F. Mizukami, S. Niwa, M. Toba, H. Ushijima and K. Itou, Appl. Chem., A: Gen. 165 (1997) 309 and references therein.
O. Vidoni, K. Philippot, C. Amiens, B. Chaudret, O. Balmes, J.O. Malm, J.O. Bovin, F. Senocq and M.J. Casanove, Angew. Chem., Int. Ed. 38 (1999) 3736; (b) K. Pelzer, O. Vidoni, K. Philippot, B. Chaudret and V. Colliere, Adv. Funct. Mater. 13 (2003) 118; (c) W. Yu, M. Liu, H. Liu, X. Ma and Z. Liu, J. Colloid Interface Sci. 208 (1998) 439; (d) M. Liu, W. Yu and H. Liu, J. Mol. Catal., A: Chem. 138 (1999) 295; (e) A. Miyazaki, K. Takeschita, K. Aika and Y. Nakano, Chem. Lett. (1998) 361; (f) Y. Wang, J. Ren, K. Deng, L. Gui and Y. Tang, Chem. Mater. 12 (2000) 1622; (g) H. Hirai, Y. Nakao and T. Toshima, J. Macromol. Sci. Chem., A 13 (1979) 727; (h) L.N. Lewis and L. Lewis, Chem. Mater. 1 (1989) 106; (i) A. Duteil, R. Quéau, B. Chaudret, R. Mazel, C. Roucau and J. S. Bradley, Chem. Mater. 5 (1993) 341; (j) H. Bönnemann, W. Brijoux, R. Brinkmann, R. Fretzen, T. Jounsen, T. Köppler, P. Neitteler and J. Richter, J. Mol. Catal. 86 (1994) 129; (l) J.-M. Planeix, N. Coustel, B. Coq, V. Brotons, P.S. Kumbhar, R. Dutartre, P. Geneste, P. Bernier and P.M. Ajayan, J. Am. Chem. Soc. 116 (1994) 7935; (m) W. Tu and H. Liu, Mater. Chem. 10 (2000) 2207; (n) S. Gao, J. Zhang, Y.-F. Zhu and C.-M. Che, New J. Chem. 24 (2000) 739; (o) G. Viau, P. Toneguzzo, A. Pierrard, O. Acher, F. Fiévet-Vincent and F. Fiévet, Scr. Mater. 44 (2001) 2263; (p) A. Spitaleri, P. Pertici, N. Scalera, G. Vitulli, M. Hoang, T.W. Turney and M. Gleria, Inorg. Chim. Acta (2003) in press.
J. Dupont, G.S. Fonseca, A.P. Umpierre, P.F.P. Fichtner and S.R. Texeira, J. Am. Chem. Soc. 124 (2002) 4228; (b) G.S. Fonseca, A.P. Umpierre, J. Dupont, P.F.P Fichtner and S.R. Texeira, Chem. Eur. J. 14 (2003) 3263; (c) C.W. Scheeren, G. Machado, J. Dupont, P.F.P. Fichtner, S.R. Texeira, Inorg. Chem. 42 (2003) 4738.
J. Dupont, P.A.Z. Suarez, R.F. de Souza, R.A. Burrow and J.P. Kintzinger, Chem. Eur. J. 6 (2000) 2377; (b) J.D. Wadhawan, U. Schroder, A. Neudeck, S.J. Wilkins, R.G. Compton, F. Marken, C.S. Consorti, R.F. de Souza and J. Dupont, J. Eletroanal. Chem. 493 (2000) 75; (c) U. Schroder, J.D. Wadhawan, R.G. Compton, F. Marken, P.A.Z. Suarez, C.S. Consorti, R.F. de Souza and J. Dupont, New J. Chem. 24 (2000) 1009.
For a recent review about ionic liquids see: J. Dupont, R.F. de Souza and P.A.Z. Suarez, Chem. Rev. 102 (2002) 3667.
For hydrogenation reactions promoted by transition-metal complexes in imidazolium ionic liquids see: (a) P.G. Jessop, R.R. Stanley, R.A. Brown, C.A. Eckert, C.L. Liotta, T.T. Ngo and P. Pollet, Green. Chem. 5 (2003) 123; (b) J. Navarro, M. Sagi, E. Sola, F.J. Lahoz, I.T. Dobrinovitch, A. Katho, F. Joo and L.A. Oro, Adv. Synth. Catal. 345 (2003) 280; (c) C.S. Consorti, A.P. Umpierre, R.F. de Souza, J. Dupont and P.A.Z. Suarez, J. Braz. Chem. Soc. 14(2003) 401; (d) C.J. Boxwell, P.J. Dyson, D.J. Ellis and T. Welton, J. Am. Chem. Soc. 124 (2002) 9334; (e) P.J. Dyson, Appl. Organomet. Chem. 16 (2002) 495; (f) C.P. Mehnert, E.J. Mozeleski and R.A. Cook, Chem. Commun. 3010 (2002); (g) R.A. Brown, P. Pollet, E. McKoon, C.A. Eckert, C.L. Liotta and P.G. Jessop, J. Am. Chem. Soc. 123 (2001) 1254; (h) S. Guernik, A. Wolfson, M. Herskowitz, N. Greenspoon and S. Geresh, Chem. Commun. (2001) 2314; (i) A. Berger, R.F. de Souza, M.R. Delgado and J. Dupont, Tetrahedron: Asymmetry 12 (2001) 1825; (j) J. Dupont, P.A.Z. Suarez, A.P. Umpierre and R.F. de Souza, Catal. Lett. 73 (2001) 211; (l) P.J. Dyson, D.J. Ellis and T. Welton, Can. J. Chem. 79 (2001) 705; (m) S. Steines, P. Wasserscheid and B.J. Driessen-Holscher, Prakt. Chem. 342 (2000) 348; (n) A.L. Monteiro, F.K. Zinn, R.F. de Souza and J. Dupont, Tetrahedron: Asymmetry 8 (1997) 177; (o) P.A.Z. Suarez, J.E.L. Dullius, S. Einloft, R.F. de Souza and J. Dupont, Inorg. Chim. Acta 255 (1997) 207; (p) Y. Chauvin, L. Mussmann and H. Olivier, Angew. Chem., Int. Ed. Engl. 34 (1996) 2698.
P.A.Z. Suarez, J.E.L. Dullius, S. Einloft, R.F. de Souza and J. Dupont, Polyhedron 15 (1996) 1217; (b) J. Dupont, P.A.Z. Suarez, C.S. Consorti and R.F. de Souza, Org. Synth. 79 (2002) 236.
B.K. Sweeny and D.G. Peters, Electrochem. Commun. 3 (2001) 712.
For a detailed discussion of CS2 and Hg(0) poisoning tests see: (a) K.S. Weddle, J.D. Aiken and R.G. Finke, J. Am. Chem. Soc. 120 (1998) 5653; (b) B.J. Hornstein, J.D. Aiken and R.G. Finke, Inorg. Chem. 41 (2002) 1625.
For the physical-chemical properties of imidazolium ionic liquids see: (a) P.A.Z. Suarez, S. Einloft, J.E.L. Dullius, R.F. de Souza and J. Dupont, J. Chim. Phys. Phys.-Chim. Biol. 95 (1998) 1626; (b) J.E.L. Dullius, P.A.Z. Suarez, S. Einloft, R.F. de Souza, J. Dupont, J. Fischer and A. De Cian, Organometallics 17 (1998) 815.
R.G. Finke, in Transition-Metal Nanoclusters, D.L. Feldheim and C.A. Foss Jr. (eds), Chapter. 2 (Marcel Dekker, New York, 2002) pp. 17-54.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rossi, L.M., Machado, G., Fichtner, P.F.P. et al. On the Use of Ruthenium Dioxide in 1-n-Butyl-3-Methylimidazolium Ionic Liquids as Catalyst Precursor for Hydrogenation Reactions. Catalysis Letters 92, 149–155 (2004). https://doi.org/10.1023/B:CATL.0000014337.40179.4a
Issue Date:
DOI: https://doi.org/10.1023/B:CATL.0000014337.40179.4a