Abstract
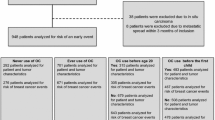
Objective: We examined breast cancer risk related to lifetime exposure to oral contraceptives (OCs) and hormone replacement therapy (HRT) in postmenopausal women. Methods: The Women's Contraceptive and Reproductive Experiences (CARE) Study was a population-based case–control study that included 1847 postmenopausal women with incident invasive breast cancer, and 1932 control subjects, identified using random digit dialing. Results: 45% of cases and 49% of controls used both OCs and HRT. OC users were not at increased risk regardless of subsequent HRT exposure. HRT users who had used OCs previously did not have a higher risk of breast cancer than women with no exposure to OCs. We observed a negative interaction (p-value: 0.032) of combined hormone replacement therapy (CHRT) and past OC use. The increase in risk with CHRT was stronger in women who had never used OCs in the past (odds ratio: 1.05; 95% confidence interval: 1.01–1.10 per year of exclusive CHRT use) than in women who had used OCs (odds ratio: 1.00; 95% confidence interval: 0.97–1.03). Conclusions: We found no indication that adverse effects of exposure to OCs or HRT appeared only in the presence of the other hormone or were exacerbated by exposure to the other hormone.
Similar content being viewed by others
References
Collaborative Group on Hormonal Factors in Breast Cancer (1996) Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53, 297 women with breast cancer and 100,239 women without breast cancer from 54 epidemiological studies. Lancet 347: 1713-1727.
Collaborative Group on Hormonal Factors in Breast Cancer (1997) Breast cancer and hormone replacement therapy: collaborative reanalysis of data from 51 epidemiological studies of 52,705 women with breast cancer and 108, 411 women without breast cancer. Lancet 350: 1047-1059.
Ross R,Paganini-Hill A,Wan P,Pike M (2000) Effect of hormone replacement therapy on breast cancer risk: estrogen versus estrogen plus progesterone. J Natl Cancer Inst 92: 323-332.
Persson I,Weiderpass E,Bergkvist L,Bergstrom R,Schairer C (1999) Risk of breast and endometrial cancer after estrogen and estrogen-progestin replacement. Cancer Causes Control 10: 253-260.
Magnusson C,Baron J,Correia N,Bergstrom R,Adami H,Persson I (1999) Breast cancer risk following long-term oestrogen and oestrogen-progestin replacement therapy. Int J Cancer 81: 339-344.
Schairer C,Lubin J,Troisi R,Sturgeon S,Brinton L,Hoover R (2000) Menopausal estrogen and estrogen-progestin replacement therapy and breast cancer risk. JAMA 283: 485-491.
Colditz G,Rosner B (2000) Cumulative risk of breast cancer to age 70 years according to risk factor status: data from the Nurses' Health Study. Am J Epidemiol 152: 950-964.
Hulley S,Furnberg C,Barrett-Conner E, et al. (2002) Noncardiovascular disease outcomes during 6.8 years of hormone therapy. Heart and Estrogen/Progestin Replacement Study Follow-up (HERS II). JAMA 288: 58-66.
Newcomb PA,Titus-Ernstoff L,Egan KM, et al. (2002) Postmenopausal estrogen and progestin use in relation to breast cancer risk. Cancer Epidemiol Biomar 11: 593-600.
Writing Group for the Women's Health Initiative Investigators (2002) Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA 288: 321-333.
Brinton LA,Brogan DR,Coates R,Swanson C,Potischman N,Stanford JL (1998) Breast cancer risk among women under 55 years of age by joint effects of usage of oral contraceptives and hormone replacement therapy. Menopause 5: 145-151.
Mills PK,Beeson WL,Phillips RL,Fraser G (1989) Prospective study of exogenous hormone use and breast cancer in Seventh-day Adventists. Cancer 64: 591-597.
Schuurman AG,van den Brandt P,Goldbohm RA (1995) Exogenous hormone use and the risk of postmenopausal breast cancer: results from the Netherlands Cohort Study. Cancer Causes Control 6: 416-424.
Stanford JL,Brinton LA,Hoover RN (1989) Oral contraceptives and breast cancer: results from an expanded case-control study. Br J Cancer 60: 375-381.
Stanford JL,Weiss NS,Voigt LF,Daling JR,Habel LA,Rossing MA (1995) Combined estrogen and progestin hormone replacement therapy in relation to risk of breast cancer in middle-aged women. JAMA 274: 137-142.
Ursin G,Tseng C-C,Paganini-Hill A, et al. (2002) Does menopausal hormone replacement therapy interact with known factors to increase risk of breast cancer. J Clin Oncol 20: 699-706.
Clemons M,Goss P (2001) Estrogen and the risk of breast cancer. N Engl J Med 344: 276-285.
Committee on the Relationship Between Oral Contraceptives and Breast Cancer (1991) Oral Contraceptives and Breast Cancer. Washington, D.C.: National Academy Press.
Hofseth L,Raafat A,Osuch J,Pathak D,Slomski C,Haslam S (1999) Hormone replacement therapy with estrogen or estrogen plus medroxyprogesterone acetate is associated with increased epithelial proliferation in the normal post-menopausal breast. J Clin Endocrinol Metab 84: 4559-4565.
Raafat AM,Hofseth LJ,Haslam SZ (2001) Proliferative effects of combination estrogen and progesterone replacement therapy on the normal postmenopausal mammary gland in a murine model. Am J Obstet Gynecol 184: 340-349.
Raafat AM,Songjiang L,Bennett JM,Hafseth LJ,Haslam SZ (2001) Estrogen and estrogen plus progestin act directly on the mammary gland to increase proliferation in a postmenopausal mouse model. J Cell Physiol 187: 81-89.
Marchbanks PA,McDonald JA,Wilson HG, et al. (2002) Oral contraceptives and the risk of breast cancer. N Engl J Med 346: 2025-2032.
Weiss LK,Burkman RT,Cushing-Haugen K, et al. (2002) Hormone replacement therapy regimens and breast cancer risk. Obstet Gynecol 100: 1148-1158.
Daling JR,Malone KE,Doody DR, et al. (2002) Relation of regimens of combined hormone replacement therapy to lobular, ductal, and other histologic types of breast carcinoma. Cancer 95: 2455-2464.
Marchbanks PA,McDonald JA,Wilson HG, et al. (2002) The NICHD Women's Contraceptive and Reproductive Experiences Study: Methods and Operational Results. Ann Epidemiol 12: 213-221.
Wingo PA,Ory HW,Layde PM, et al. (1998) The evaluation of the data collection process for a multicenter, population-based case-control design. Am J Epidemiol 128: 206-217.
West S,Strom B (2000) Validity of Pharmacoepidemiology Drug and Diagnosis Data. In: Strom B, ed. Pharmacoepidemiology, 3rd ed. Sussex: John Wiley.
Pike MC,Ross RK,Spicer DV (1998) Problems involved in including women with simple hysterectomy in epidemiologic studies measuring the effects of hormone replacement therapy on breast cancer risk. Am J Epidemiol 147: 718-721.
Rockhill B,Colditz GA,Rosner B (2000) Bias in breast cancer analyses due to error in age at menopause. Am J Epidemiol 151: 404-408.
Malone KE,Daling JR,Weiss NS (1993) Oral contraceptives in relation to breast cancer. Epidemiol Rev 15: 80-97.
U.S. Preventive Services Task Force (2002) Postmenopausal hormone replacement therapy for primary prevention of chronic conditions: recommendations and rationale. Ann Intern Med 137: 834-839.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Norman, S.A., Berlin, J.A., Weber, A.L. et al. Combined effect of oral contraceptive use and hormone replacement therapy on breast cancer risk in postmenopausal women. Cancer Causes Control 14, 933–943 (2003). https://doi.org/10.1023/B:CACO.0000007967.25865.29
Issue Date:
DOI: https://doi.org/10.1023/B:CACO.0000007967.25865.29