Abstract
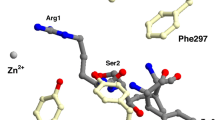
A comparative study of secondary specificities of enteropeptidase and trypsin was performed using peptide substrates with general formula A-(Asp/Glu) n -Lys(Arg)-↓-B, where n = 1-4. This was the first study to demonstrate that, similar to other serine proteases, enteropeptidase has an extended secondary binding site interacting with 6-7 amino acid residues surrounding the peptide bond to be hydrolyzed. However, in the case of typical enteropeptidase substrates containing four negatively charged Asp/Glu residues at positions P2-P5, electrostatic interaction between these residues and the secondary site Lys99 of the enteropeptidase light chain is the main factor that determines hydrolysis efficiency. The secondary specificity of enteropeptidase differs from the secondary specificity of trypsin. The chromophoric synthetic enteropeptidase substrate G5DK-F(NO2)G (k cat/K m = 2380 mM–1·min–1) is more efficient than the fusion protein PrAD4K-P26 (k cat/K m = 1260 mM–1·min–1).
Similar content being viewed by others
REFERENCES
Kunitz, M. (1939) J. Gen. Physiol., 22, 429–446.
Sharma, A., Khoury-Christianson, A. M., White, S. P., Dhanjal, N. K., Huang, W., Paulhiac, C., Friedman, E. J., Manjula, B. N., and Kumar, R. (1994) Proc. Natl. Acad. Sci. USA, 91, 9337–9341.
Uegaki, K., Nemoto, N., Shimizu, M., Wada, T., Kyogoku, Y., and Kobayashi, Y. (1996) FEBS Lett., 379, 47–50.
Safi, W., Maiorano, J. N., and Davidson, W. S. (2001) J. Lipid Res., 42, 864–872.
Agnihotri, R., Crawford, H. C., Haro, H., Matrisian, L. M., Havrda, M. C., and Liaw, L.(2001) J. Biol. Chem., 276, 28261–28267.
Hosfield, T., and Lu, Q. (1999) Analyt. Biochem., 269, 10–16.
Mikhailova, A. G., and Rumsh, L. D. (2000) Appl. Biochem. Biotechnol., 88, 159–174.
Lu, D., Futterer, K., Korolev, S., Zheng, X., Tan, K., Waksman, G., and Sadler, J. E. (1999) J. Mol. Biol., 292, 361–373.
Mikhailova, A. G., and Rumsh, L. D. (1998) Bioorg. Khim., 24, 282–287.
Mikhailova, A. G., and Rumsh, L. D. (1999) FEBS Lett., 442, 226–230.
Lu, D., and Sadler, J. E. (1998) in Handbook of Proteolytic Enzymes (Barret, A. J., Rawlings, N. D., and Woessner, J. F., eds.) Academic Press, London, pp. 50–54.
Likhareva, V. V., Mikhailova, A. G., and Rumsh, L. D. (2002) Vopr. Med. Khim., 48, 561–569.
Kapust, R. B., Tozser, J., Fox, J. D., Anderson, D. E., Cherry, S., Copeland, T. D., and Waugh, D. S. (2001) Protein Eng., 14, 993–1000.
Mutt, V., Tatemoto, K., Carlquist, M., and Light, A. (1981) Biosci. Rep., 1, 651–659.
Likhareva, V. V., Vaskovsky, V. B., Shepel', N. E., Garanin, S. K., Mikhailova, A. G., and Rumsh, L. D. (2003) Bioorg. Khim., 29, 129–134.
Likhareva, V. V., Mikhailova, A. G., Vaskovsky, B. V., Garanin, S. K., and Rumsh, L. D. (2002) Lett. Peptide Sci., 9, 71–76.
Hofmann, Th., and Hodges, R. S. (1982) Biochem. J., 203, 603–610.
Mikhailova, A. G., Vorotyntseva, T. I., Bessmertnaya, L. Ya., and Antonov, V. K. (1984) Biokhimiya, 49, 1483–1487.
Eisenthal, R., and Cornish-Bowden, A. (1974) Biochem. J., 139, 715–720.
Craik, C. S., Largman, C., Fletcher, T., Roczniak, S., Barr, P. J., Fletterick, R., and Rutter, W. J. (1985) Science, 222, 291–297.
Grant, D. A. V., and Hermon-Taylor, J. (1979) Biochim. Biophys. Acta, 567, 207–215.
Baratty, J., and Maroux, S. (1976) Biochim. Biophys. Acta, 452, 488–496.
Lu, D., Yuan, X., Zheng, X., and Sadler, J. E. (1997) J. Biol. Chem., 272, 31293–31300.
Abita, J. P., Delaage, M., and Lazdunski, M. (1969) Eur. J. Biochem., 8, 314–324.
Hedstrom, L. (2002) Chem. Rev., 102, 4501–4523.
Bianchini, E. P., Louvain, V. B., Marque, P.-E., Juliano, M. A., Juliano, L., and Le Bonniec, B. F. (2002) J. Biol. Chem., 277, 20527–20534.
Fiedler, F. (1987) Eur. J. Biochem., 163, 303–312.
Pozsgay, M., Cs. SzabÓ, G., Bajusz, S., Simonsson, R., Gáspár, R., and Elodi, P. (1981) Eur. J. Biochem., 115, 497–502.
Kurth, T., Grahn, S., Thormann, M., Ullman, D., Hofmann, H.-J., Jakubke, H.-D., and Hedstrom, L. (1998) Biochemistry, 37, 11434–11440.
Kurth, T., Ullman, D., Jakubke, H.-D., and Hedstrom, L. (1997) Biochemistry, 36, 10098–10104.
Schellenberger, V., Turck, C. W., and Rutter, W. J. (1994) Biochemistry, 33, 4251–4257.
Grahn, S., Kurth, T., Ullman, D., and Jakubke, H.-D. (1999) Biochim. Biophys. Acta, 1431, 329–337.
Reyda, S., Sohn, Ch., Kiebe, G., Rall, K., Ullman, D., Jakubke, H.-D., and Stubbs, M. T. (2003) J. Mol. Biol., 325, 963–977.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mikhailova, A.G., Likhareva, V.V., Vaskovsky, B.V. et al. Study of Secondary Specificity of Enteropeptidase in Comparison with Trypsin. Biochemistry (Moscow) 69, 909–917 (2004). https://doi.org/10.1023/B:BIRY.0000040224.47278.3b
Issue Date:
DOI: https://doi.org/10.1023/B:BIRY.0000040224.47278.3b