Abstract
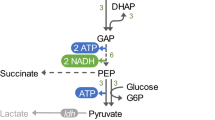
The thermophilic bacterium, Moorella sp. HUC22-1, newly isolated from a mud sample, produced ethanol from H2 and CO2 during growth at 55 °C. In batch cultures in serum bottles, 1.5 mm ethanol was produced from 270 mm H2 and 130 mm CO2 after 156 h, whereas less than 1 mm ethanol was produced from 23 mm fructose after 33 h. Alcohol dehydrogenase and acetaldehyde dehydrogenase activities were higher in cells grown with H2 and CO2 than those grown with fructose. The NADH/NAD+ and NADPH/NADP+ ratios in cells grown with H2 and CO2 were also higher than those in cells grown with fructose. When the culture pH was controlled at 5 with H2 and CO2 in a fermenter, ethanol production was 3.7-fold higher than that in a pH-uncontrolled culture after 220 h.
Similar content being viewed by others
References
Abrini J, Naveau H, Nyns EJ (1994) Clostridium autoethanogenum, sp. nov., an anaerobic bacterium that produces ethanol from carbon monoxide. Arch. Microbiol. 161: 345–351.
Andreesen JR, Schaupp A, Neurauter C, Brown A, Ljungdahl LG (1973) Fermentation of glucose, fructose, and xylose by Clostridium thermoaceticum: effect of metals on growth yield, enzymes, and the synthesis of acetate from CO 2. J. Bacteriol. 114: 743–751.
Barrow GI, Feltham RKA (1993) Cowan and Steel's Manual for the Identification of Medical Bacteria, 3rd edn. Cambridge: Cambridge University Press, pp. 21–42.
Berríos-Rivera SJ, Bennett GN, San K-Y (2002) Metabolic engineering of Escherichia coli: increase of NADH availability by overexpressing an NAD +-dependent formate dehydrogenase. Metabolic Eng. 4: 217–229.
Buschhorn H, Dürre P, Gottschalk G (1989) Production and utilization of ethanol by the homoacetogen Acetobacterium woodii. Appl. Environ. Microbiol. 55: 1835–1840.
Drake HL, Küsel K, Matthies C (2002) Ecological consequences of the phylogenetic and physiological diversities of acetogens. Antonie van Leeuwenhoek 81: 203–213.
Fröstl JM, Seifritz C, Drake HL (1996) Effect of nitrate on the auto-trophic metabolism of the acetogens Clostridium thermoauto-trophicum and Clostridium thermoaceticum. J. Bacteriol. 178: 4597–4603.
Grethlein AJ, Worden RM, Jain MK, Datta R (1991) Evidence for production of n-butanol from carbon monoxide by Butyribacterium methylotrophicum. J. Ferment. Bioeng. 72: 58–60.
Hungate RE (1969) A roll tube method for cultivation of strict anaerobes. Meth. Microbiol. 3B: 117–132.
Jones DT, Woods DR (1986) Acetone-butanol fermentation revisited. Microbiol. Rev. 50: 484–524.
Klingenberg M (1985) Metabolites 2: tri-and dicarboxylic acids, purines, pyrimidines and derivatives, coenzymes, inorganic compounds. In: Bergmeyer HU, ed. Methods of Enzymatic Analysis, Vol. 7. New York, London: Academic Press, pp. 251–267.
Lovitt RW, Shen GJ, Zeikus JG (1988) Ethanol production by thermophilic bacteria: biochemical basis for ethanol and hydrogen tolerance in Clostridium thermohydrosulfuricum. J. Bacteriol. 170: 2809–2815.
Marwoto B, Nakashimada Y, Kakizono T, Nishio N (2002) Enhancement of (R,R)-2,3-butanediol production from xylose by Paenibacillus polymyxa at elevated temperatures. Biotechnol. Lett. 24: 109–114.
Marwoto B, Nakashimada Y, Kakizono T, Nishio N (2004) Metabolic analysis of acetate accumulation during xylose consumption by Paenibacillus polymyxa. Appl. Microbiol. Biotechnol. 64: 112–119.
Miller TL, Wolin MJ (1974) A serum bottle modification of the Hungate technique for cultivating obligate anaerobes. Appl. Microbiol. 27: 985–987.
Nakashimada Y, Rachman MA, Kakizono T, Nishio N (2002) Hydrogen production of Enterobacter aerogenes altered by extracellular and intracellular redox states. Int. J. Hydrogen Energ. 27: 1399–1405.
Nishio N, Eguchi SY, Kawashima H, Nagai S (1983) Mutual conversion between H 2 plus CO 2 and formate by a formate-utilizing methanogen. J. Ferment. Technol. 61: 557–561.
Simon H, White H, Lebertz H, Thanos I (1987) Reduction of 2-enoates and alkanoates with carbon monoxide or formate, viologens, and Clostridium thermoaceticum to saturated acids and unsaturated alcohols. Angew. Chem. Int. Ed. Engl. 26: 785–787.
Tanner RS, Miller LM, Yang D (1993) Clostridium ljugdahlii sp. nov., an acetogenic species in clostridial rRNA homology group I. Int. J. Sys. Bacteriol. 43: 232–236.
Vasconcelos I, Girbal L, Soucaille P (1994) Regulation of carbon and electron flow in Clostridium acetobutylicum grown in chemostat culture at neutral pH on mixtures of glucose and glycerol. J. Bacteriol. 176: 1443–1450.
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 173: 697–703.
White H, Lebertz H, Thanos I, Simon H (1987) Clostridium thermo-aceticum forms methanol from carbon monoxide in the presence of viologen dyes. FEMS Microbiol. Lett. 43: 173–176.
Wimpenny JWT, Firth A (1972) Levels of nicotinamide adenine dinucleotide and reduced nicotinamide adenine dinucleotide in facultative bacteria and the effect of oxygen. J. Bacteriol. 111: 24–32.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sakai, S., Nakashimada, Y., Yoshimoto, H. et al. Ethanol production from H2 and CO2 by a newly isolated thermophilic bacterium, Moorella sp. HUC22-1. Biotechnology Letters 26, 1607–1612 (2004). https://doi.org/10.1023/B:BILE.0000045661.03366.f2
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000045661.03366.f2