Abstract
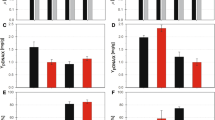
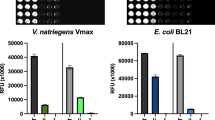
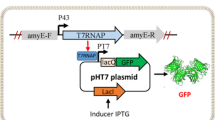
Co-expression of Vitreoscilla hemoglobin (VHb) can enhance production of foreign proteins in several microorganisms, including Escherichia coli. Production of foreign proteins [green fluorescent protein (GFP) and organophosphorous hydrolase (OPH)] has been examined in two typical industrial E. coli strains, W3110 (a K12 derivative) and BL21 (a B derivative). In particular, we investigated the effects of VHb co-expression and media glucose concentration on target protein production. We employed the nar O2-dependent promoter for self-tuning of VHb expression based on the natural changes in dissolved O2 levels over the duration of culture. Foreign protein production in strain BL21 was decreased by a high glucose concentration but co-expression of VHb had no effect on this. In contrast, co-expression of VHb in strain W3110 overrode the glucose-induced repression and resulted in steady expression of foreign proteins.
Similar content being viewed by others
References
Akesson M, Hagander P, Axelsson JP (2001) Avoiding acetate accumulation in Escherichia colicultures using feedback control of glucose feeding. Biotechnol. Bioeng. 73: 223–230.
Bauer KA, Bassat AB, Sawson M, Puente VT, Neway JO (1990) Improved expression of human interleukin-2 in high-cell-density fermentor cultures of Escherichia coliK-12 by a phosphotransacetylase mutant. Appl. Environ. Microbiol. 56: 1296–1302.
Brosius J, Erfle M, Storella J (1985) Spacing of the -10 and -35 regions in the tacpromoter: effect on its in vivoactivity. J. Biol. Chem. 260: 3539–3541.
Caldwell SR, Newcomb JR, Schlecht KA, Raushel FM (1991) Limits of diffusion in the hydrolysis of substrates by the phophotriesterase from Pseudomonas diminuta. Biochem. 30: 7438–7444.
Cha HJ, Wu CF, Valdes JJ, Rao G, Bentley WE (2000) Observations of green fluorescent protein as a fusion partner in genetically engineered Escherichia coli: monitoring protein expression and solubility. Biotechnol. Bioeng. 67: 565–574.
de Boer HA, Comstock LJ, Vasser M (1983) The tacpromoter: a functional hybrid derived from the trpand lacpromoters. Proc. Nat. Acad. Sci. USA 80: 21–25.
Dikshit KL, Webster DA (1988) Cloning, characterization and expression of the bacterial hemoglobin gene from Vitreoscillain Escherichia coli. Gene 70: 377–386.
Donarski WJ, Dumas DP, Heitmeyer DP, Lewis VE, Raushel FM (1989) Structure-activity relationship in the hydrolysis of substrates by the phosphotriesterase from Pseudomonas diminuta. Biochem. 28: 4650–4655.
Enayati N, Tari C, Parulekar SJ, Stark BC, Webster DA (1999) Production of a-amylase in fed-batch cultures of vgb+ and vgb- recombinant Escherichia coli: some observations. Biotechnol. Prog. 15: 640–645.
Grimsley JK, Scholtz JM, Pace CN, Wild JR (1997) Organophosphorus hydrolase is a remarkably stable enzyme that unfolds through a homodimeric intermediate. Biochem. 36: 14366–14374.
Hahm DH, Pan JG, Rhee JS (1994) Characterization and evaluation of a pta(phosphotransacetylase) negative mutant of Escherichia coliHB101 as production host of foreign lipase. Biotechnol. Bioeng. 42: 100–107.
Han SJ, Chang HN, Lee J (1998) Fed-batch cultivation of an oxygen-dependent inducible promoter system, the narpromoter in Escherichia coliwith an inactivated naroperon. Biotechnol. Bioeng. 59: 400–406.
Holms WH (1986) The central metabolic pathways in Eschericchia coli: relationship between flux and control at a branchpoint, efficiency of conversion to biomass, and excretion of acetate. Curr. Topic. Cell. Regul. 28: 69–105.
Kallio PT, Bailey JE (1996) Intracellular expression of Vitreoscillahemoglobin (VHb) enhances total protein secretion and improves the production of a-amylase and neutral protease in Bacillus subtilis. Biotechnol. Prog. 12: 31–39.
Kang DG, Kim JYH, Cha HJ (2002a) Enhanced detoxification of organophosphates using recombinant Escherichia coliwith coexpression of organophosphorus hydrolase and bacterial hemoglobin. Biotechnol. Lett. 24: 879–883.
Kang DG, Kim YK, Cha HJ (2002b) Comparison of green fluorescent protein expression in two industrial Escherichia colistrains, BL21 and W3110, under co-expression of bacterial hemoglobin. Appl. Microbiol. Biotechnol. 59: 523–528.
Khosla C, Bailey JE (1988) Heterologous expression of a bacterial hemoglobin improves the growth properties of recombinant Escherichia coli. Nature 331: 633–635.
Lee J, Cho MH, Lee J (1996) Characterization of an oxygendependent inducible promoter system, the narpromoter, and Escherichia coliwith an inactivated naroperon. Biotechnol. Bioeng. 52: 572–578.
Li SF, DeMoss JA (1988) Location of sequences in the narpromoter of Escherichia colirequired for regulation by Fnr and NarL. J. Biol. Chem. 263: 13700–13705.
Majewski RA, Domach MM (1990) Simple constrainedoptimization view of acetate overflow in E. coli. Biotechnol. Bioeng. 35: 732–738.
Robbins JW, Taylor KB (1989) Optimization of Escherichia coligrowth by controlled addition of glucose. Biotechnol. Bioeng. 34: 1289–1294.
Shiloach J, Kaufman J, Guillard AS, Fass R (1996) Effect of glucose supply strategy on acetate accumulation, growth, and recombinant protein production by Escherichia coliBL21(?DE3) and Escherichia coliJM109. Biotechnol. Bioeng. 49: 421–428.
van de Walle M, Shiloach J (1998) Proposed mechanism of acetate accumulation in two recombinant Escherichia colistrains during high density fermentation. Biotechnol. Bioeng. 57: 71–78.
Wu CF, Cha HJ, Rao G, Valdes JJ, Bentley WE (2000) A green fluorescent protein fusion strategy for monitoring the expression, cellular location, and separation of biologically active organophosphorus hydrolase. Appl. Microbiol. Biotechnol. 54: 78–83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, Y.K., Kang, D.G., Choi, SS. et al. Co-expression of bacterial hemoglobin overrides high glucose-induced repression of foreign protein expression in Escherichia coli W3110. Biotechnology Letters 26, 1173–1178 (2004). https://doi.org/10.1023/B:BILE.0000035491.64912.84
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000035491.64912.84