Abstract
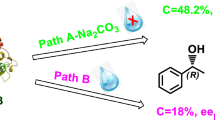
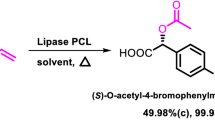
The ionic liquid, 1-butyl-3-methylimidazolium bis[(trifluoromethyl)sulfonyl]amide ([Bmim] [NTf2]), was used as a reaction medium for the kinetic resolution of rac-2-pentanol catalyzed by free Candida antarctica lipase B, using vinyl propionate at 2% (v/v) water content. The synthetic activity of lipase in [Bmim] [NTf2] was up 2.5-times greater than in hexane, and showed high enantioselectivity (ee > 99.99%). The optimal temperature and pH were 60 °C and 7, respectively. A decrease in water activity (aw) produced a decay in synthetic activity, and an exponential increase in selectivity.
Similar content being viewed by others
References
Berberich JA, Kaar JL, Russell AJ (2003) Use salt hydrates pairs to control water activity for enzyme catalysis in ionic liquids. Biotechnol. Prog. 19: 1029–1032
Cao L, van Langen L, Sheldon RA (2003) Immobilised enzymes: carrier-bound or carrier free? Curr. Opin. Biotechnol. 14: 1–8.
Dupont J, de Souza R, Suarez PAZ (2002) Ionic liquids (molten salts) phase organometallic catalysis. Chem. Rev. 102: 3667–3692.
Dzyuba SV, Bartsch RA (2003) Recent advances in applications of room-temperature ionic liquid/supercritical CO2 systems. Angew. Chem. Int. Ed. 42: 148–150.
Eckstein M, Wasserscheid P, Kragl U (2002a) Enhanced enantioselectivity of lipase from Pseudomonas sp. at high temperatures and fixed water activity in the ionic liquid, 1-butyl-3-methylimidazolium bis[(trifluoromethyl)sulfonyl]amide. Biotechnol. Lett. 24: 763–767.
Eckstein M, Sesing M, Kragl U, Adlercreutz P (2002b) At low water activity á-chymotrypsin is more active in an ionic liquid than in non-ionic organic solvents. Biotechnol. Lett. 24: 867–872.
Klivanov AM (2001) Improving enzymes by using them in organic solvents. Nature 409: 241–246.
Lozano P, de Diego T, Carrié D, Vaultier M, Iborra JL (2001) Overstabilization of Candida antarctica lipase B by ionic liquids in ester synthesis. Biotechnol. Lett. 23: 1529–1533.
Lozano P, De Diego T, Carrié, D, Vaultier M, Iborra JL (2002) Continuous green biocatalytic processes using ionic liquids and supercritical carbon dioxide. Chem. Commun. 692-693.
Lozano P, De Diego T, Carrié D, Vaultier M, Iborra JL (2003a) Enzymatic ester synthesis in ionic liquids. J. Mol. Catal. B Enzym. 21: 9–13.
Lozano P, De Diego T, Carrié D, Vaultier M, Iborra JL (2003b) Lipase catalysis in ionic liquids and supercritical carbon dioxide at 150 C. Biotechnol. Prog. 19: 380–382.
Ottosson J, Hult K (2001) Influence of acyl chain length on the enantioselectivity of Candida antarctica lipase B and its thermodynamic components in kinetic resolution of sec-alcohols. J. Mol. Catal. B Enzym. 11: 1025–1028.
Park S, Kazlauskas RJ (2001) Improved preparation and use of room-temperature ionic liquids in lipase-catalyzed enantio-and regioselective acylations. J. Org. Chem. 66: 8395–8401.
Patel RN (2001) Enzymatic synthesis of chiral intermediates for drug development. Adv. Synth. Catal. 343: 527–546.
Patel RN, Banerjee A, Nanduri V, Goswami A, Comezoglu FT (2000) Enzymatic resolution of racemic secondary alcoholds by lipase B from Candida antarctica. J. Am. Oil Chem. Soc. 77: 1015–1019.
Persson M, Bornscheuer UT (2003) Increased stability of an esterase from Bacillus stearothermophilus in ionic liquids as compared to organic solvents. J. Mol. Catal. B Enzym. 22: 21–27.
Stinson SC (2000) Chiral drugs. Chem. Eng. News 78: 55–78.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noël, M., Lozano, P., Vaultier, M. et al. Kinetic resolution of rac-2-pentanol catalyzed by Candida antarctica lipase B in the ionic liquid, 1-butyl-3-methylimidazolium bis[(trifluoromethyl)sulfonyl]amide. Biotechnology Letters 26, 301–306 (2004). https://doi.org/10.1023/B:BILE.0000015429.28402.50
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000015429.28402.50