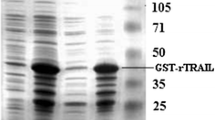
Abstract
As a new member of tumor necrosis factor (TNF) superfamily, TNF-related apoptosis-inducing ligand (Apo2L/TRAIL) was produced mainly as inclusion bodies by recombinant Escherichia coli with a temperature-inducible expression system. High concentrations of both biomass (65 g dry cells l−1) and inactive TRAIL (4.8 g l−1) were obtained by applying a high-cell-density cultivation procedure. After the inclusion bodies were washed and solubilized, TRAIL refolded when at 1 mg ml−1 by a simple pulse dilution method with a 35% yield. Renatured TRAIL was purified to electrophoretic homogeneity by one-step immobilized metal affinity chromatography. The purified TRAIL showed strong cytotoxicity activity against human pancreatic 1990 tumor cells, with ED50 about 1.6 μg ml−1.
Similar content being viewed by others
References
Aggarwal BB, Kohr WJ, Hass PE, Moffat B, Spencer SA, Henzel WJ, Bringman TS, Nedwin GE, Goeddel DV, Harkins RN (1985) Human tumor necrosis factor. J. Biol. Chem. 260: 2345–2354.
Futami J, Tsushima Y, Tada H, Seno M, Yamada H (2000) Convenient and efficient in vitro folding of disulfide-containing globular protein from crude bacterial inclusion bodies. J. Biochem. 127: 435–441.
Hevehan DL, De Bernardez Clark E (1997) Oxidative renaturation of lysozyme at high concentrations. Biotechnol. Bioeng. 54: 221–230.
Hymowitz SG, O'Connel MP, Ultsch MH, Hurst A, Totpal K, Ashkenazi A, de Vos AM, Kelley RF (2000) A unique zincbinding site revealed by a high-resolution X-ray structure of homotrimeric Apo2L/TRAIL. Biochemistry 39: 633–640.
Lee SY (1996) High cell-density culture of Escherichia coli. Trends Biotechnol. 14: 98–105.
Maachupalli-Reddy J, Kelley BD, De Bernardez Clark E (1997) Effect of inclusion body contaminants on the oxidative renaturation of hen egg white lysozyme. Biotechnol. Prog. 13: 144–150.
Rudolph R (1990) Renaturation of recombinant, disulfide-bonded proteins from ‘inclusion bodies’. In: Tschesche H, ed. Modern Methods in Protein and Nucleic Acid Analysis. Berlin, New York: Walter de Grutyer, pp. 149–171.
Rudolph R, Fischer S (1990) Process for Obtaining Renatured Proteins. U.S. Patent 4 933 434.
Schmidt M, Babu KR, Khanna N, Marten S, Rinas U (1999) Temperature-induced production of recombinant human insulin in high-cell density cultures of recombinant Escherichia coli. J. Biotechnol. 68: 71–83.
Seeger A, Schneppe B, McCarthy JEG, Deckwer WD, Rinas U (1995) Comparison of temperature-and isopropyl-?-D-thiogalacto-pyranoside-induced synthesis of basic fibroblast growth factor in high-cell-density cultures of recombinant Escherichia coli. Enzyme Microb. Technol. 17: 947–953.
Walczak H, Miller RE, Ariall K, Gliniak B, Griffith TS, Kubin M, Chin W, Jones J, Woodward A, Le T, Smith C, Smolak P, Goodwin RG, Rauch CT, Schuh JCL, Lynch DH (1999) Tumoricidal activity of tumor necrosis factor-related apoptosis-inducing ligand in vivo. Nat. Med. 5: 157–163.
Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C Smith CA Goodwin RG (1995) Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3: 673–682.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, YL., Xia, XX., Zhang, Y. et al. Refolding and purification of Apo2L/TRAIL produced as inclusion bodies in high-cell-density cultures of recombinant Escherichia coli . Biotechnology Letters 25, 2097–2101 (2003). https://doi.org/10.1023/B:BILE.0000007072.56577.0c
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000007072.56577.0c