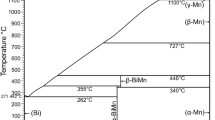
Abstract
Phase equilibria in the binary system Bi2O3-ZnO were studied by quenching technique. Heat-treated compositions were subjected to X-ray diffraction for phase identification, and differential thermal analysis, optical and scanning electron microscopy were used to determine the solid-liquid equilibria occurring in this system. The data thus obtained revealed that incorporation of a small amount of ZnO to the high-temperature face-centered cubic lattice of Bi2O3 leads to the formation of a body-centered cubic solid solution (γ-Bi2O3), which extends up to a composition of 2.2 mol% ZnO at a temperature near 750°C. On cooling, the γ-Bi2O3 solid solution undergoes a eutectoid transformation at a temperature of 710°C to yield the low-temperature monoclinic polymorph of Bi2O3 (α-Bi2O3) and Bi38ZnO58. The eutectoid occurs at a composition of 1.8 mol% ZnO. The compound Bi38ZnO58 has a crystal structure analogous to the body-centered cubic γ-Bi2O3 solid solution and melts incongruently at a temperature near 753 ± 2°C to yield γ-Bi2O3 and liquid. A binary eutectic occurs between Bi38ZnO58 and ZnO at a composition near 25 ± 1.0 mol% ZnO with a melting temperature of 738 ±2°C. Based on the data obtained in this study, a revised phase diagram of the binary system Bi2O3-ZnO is proposed.
Similar content being viewed by others
References
M. Matsuoka, Jpn. J. Appl. Phys. 10 (1971) 736.
J. Wong, J. Amer. Ceram. Soc. 57 (1974) 357.
T. K. Gupta, ibid. 73 (1990) 1817.
D. R. Clark, ibid. 82 (1999) 485.
M. F. Yan, H. C. Ling and W. W. Rhodes, ibid. 73 (1990) 1106.
H. Kagata, T. Inoue, J. Kato and I. Kameyama, Jpn. J. Appl. Phys. 31 (1992) 3152.
D. P. Cann, C. A. Randalli and T. R. Shrout, Solid State Comm. 100 (1996) 529.
D. Liu, Y. Liu, S. Huang and X. Li, J. Amer. Cearm. Soc. 76 (1993) 2129.
X. Wang, H. Wang and X. Yao, ibid. 80 (1997) 2745.
H. Wang and XI. Yao, J. Mater. Res. 16 (2001) 83.
J. C. Nino, M. T. Lanagan and C. A. Randal, J. Appl. Phys. 89 (2001) 4512.
M. Valant and P. K. Davies, J. Mater. Sci. 34 (1999) 5437.
M. Valant, J. Amer. Ceram. Soc. 83 (2000) 147.
E. M. Levin and R. S. Roth, J. Res. Natl. Bur. Std. 68A (1964) 197.
G. M. Saffronov, V. N. Batog, T. V. Stepanyuk and P. M. Federov, Russ. J. Inorg. Chem. (Engl. Transl.) 16 (1971) 460.
T. M. Bruton, O. F. Hill, P. A. C. Whiffin and J. C. Brice, J. Cryst. Growth 32 (1976) 27.
T. M. Bruton, Ferroelectrics 7 (1974) 259.
J. Wong and G. Morris, Ceram. Bull. 53 (1974) 816.
D. C. Craig and N. C. Stephenson, J. Solid State Chem. 15 (1975) 1.
S. F. Radev and V. I. Simonov, Kristallografiya 37 (1992) 914.
JCPDS Card No. 41-0253 (1989).
JCPDS Card No. 42-0138 (1991).
J. Kim, T. Kimura and T. Yamaguchi, J. Amer. Ceram. Soc. 72 (1898) 1541.
J.-H. Hwang, T. O. Mason and V. Dravid, ibid. 77 (1994) 1499.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guha, J.P., Kunej, Š. & Suvorov, D. Phase equilibrium relations in the binary system Bi2O3-ZnO. Journal of Materials Science 39, 911–918 (2004). https://doi.org/10.1023/B:JMSC.0000012921.62765.05
Issue Date:
DOI: https://doi.org/10.1023/B:JMSC.0000012921.62765.05