Abstract
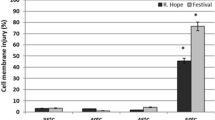
Strawberry plants (Fragaria×ananassa Duch.) cvs. Nyoho and Toyonoka were exposed to temperatures of 20, 33, and 42 °C for 4 h, and protein patterns in leaves and flowers was analyzed by 2-dimensional polyacrylamide gel electrophoresis and immunoblotting. In leaves and flowers of both cultivars, the content of most proteins decreased, but a few new proteins appeared in response to heat stress. These heat shock proteins (Hsps) were detected in the range of 19 – 29 kDa in leaves, and 16 – 26 kDa in flowers. The intensity of a 43 kDa protein spot increased in response to heat stress in Nyoho flowers, but not in Toyonoka flowers. The peaHsp17.7 antibody recognized one band at approximately 26 kDa in leaves, and two bands at approximately 16 and 17 kDa in flowers of both cultivars. These results show that the effects of heat stress on Hsp synthesis in strawberry plants differ between plant organs and between cultivars.
Similar content being viewed by others
References
Atkinson, B.G., Rauzada, M., Bouchard, R.A., Frappier, J.R.H., Walden, D.B.: The independent stage-specific expression of the 18 kDa heat shock protein genes during microsporogenesis in Zea mays L.-Dev. Genet. 14: 15-26, 1993.
Boston, R.S., Viitanen, P.V., Vierling, E.: Molecular chaperones and protein folding in plants.-Plant mol. Biol. 32: 191-222, 1996.
Civello, P.M., Martinez, G.A., Chaves, A.R., Anon, M.C.: Heat treatments delay ripening and postharvest decay of strawberry fruit.-J. agr. Food Chem. 45: 4589-4594, 1997.
Coca, M.A., Almoguera, C., Jordano, J.: Expression of sunflower low molecular weight heat shock proteins during embryogenesis and persistence after germination: localization and possible functional implications.-Plant mol. Biol. 25: 479-492, 1994.
DeRocher, A.E., Helm, K.W., Lauzon, L.M., Vierling, E.: Expression of a conserved family of cytoplasmic low molecular weight heat shock proteins during heat stress and recovery.-Plant Physiol. 96: 1038-1047, 1991.
DeRocher, A.E., Vierling, E.: Developmental control of small heat shock protein expression during pea seed maturation.-Plant J. 5: 93-102, 1994.
Fender, S.E., O'Connell, M.A.: Heat shock protein expression in thermotolerant and thermosensitive lines of cotton.-Plant Cell Rep. 8: 37-40, 1989.
Forreiter, C., Nover, L.: Heat induced stress proteins and the concept of molecular chaperones.-J. Biosci. 23: 287-302, 1998.
Fray, R.G., Lycett, G.W., Grierson, D.: Nucleotide sequence of heat shock and ripening-related cDNA from tomato.-Nucleic Acids Res. 18: 7148, 1990.
Gyorgyey, J., Gartner, A., Nemeth, K., Magyar, Z., Hirt, H., Heberle-Bors, E., Dudits, D.: Alfalfa heat shock genes are differentially expressed during somatic embryogenesis.-Plant mol. Biol. 16: 999-1007, 1991.
Hartl, F.U.: Molecular chaperones in cellular protein folding.-Nature 381: 571-580, 1996.
Howarth, C.J., Skot, K.P.: Detailed characterization of heat shock protein synthesis and induced thermotolerance in seedlings of Sorghum bicolor L.-J. exp. Bot. 45: 1353-1363, 1994.
Jorgensen, J.A., Rosenow, D.T., Nguyen, H.T.: Genotypic comparisons of heat shock protein synthesis in sorghum.-Crop Sci. 33: 638-641, 1992.
Joshi, C.P., Klueva, N.Y., Morrow, K.J., Nguyen, H.T.: Expression of a unique plastid-localized heat-shock protein is genetically linked to acquired thermotolerance in wheat.-Theor. appl. Genet. 95: 834-841, 1997.
Lafayette, P.R., Nagao, R.T., O'Grady, K., Vierling, E., Key, J.L.: Molecular characterization of cDNAs encoding low-molecular-weight heat shock proteins of soybean.-Plant mol. Biol. 30: 159-169, 1996.
Lee, G.J., Pokala, N., Vierling, E.: Structure and in vitro molecular chaperone activity of cytosolic small heat shock proteins from pea.-J. biol. Chem. 270: 10432-10438, 1995.
Lenne, C., Block, M.A., Garin, J., Douce, R.: Sequence and expression of the mRNA encoding HSP22, the mitochondrial small heat-shock protein in pea leaves.-Biochem. J. 311: 805-813, 1995.
Lindquist, S.: The heat shock response.-Annu. Rev. Biochem. 55: 1151-1191, 1986.
Malik, M.K., Slovin, J.P., Hwang, C.H., Zimmerman, J.L.: Modified expression of a carrot small heat shock protein gene, HSP17.7, results in increased or decreased thermotolerance.-Plant J. 20: 88-99, 1999.
Mascarenhas, J.P., Crone, D.E.: Pollen and the heat shock response.-Sex. Plant Reprod. 9: 370-374, 1996.
Medina-Escobar, N., Cardenas, J., Munoz-Blanco, J., Caballero, J.L.: Cloning and molecular characterization of a strawberry fruit ripening-related cDNA corresponding a mRNA for a low-molecular-weight heat shock protein.-Plant mol. Biol. 36: 33-42, 1998.
Park, S.Y., Shivaji, R., Krans, J.V., Luthe, D.S.: Heat-shock response in heat-tolerant and nontolerant variants of Agrostis palustris Huds.-Plant Physiol. 111: 515-524, 1996.
Parsell, D.A., Lindquist, S.: The function of heat-shock proteins in stress-tolerance: degradation and reactivation of proteins.-Annu. Rev. Genet. 27: 437-496, 1993.
Preczewski, P.J., Heckathorn, S.A., Downs, C.A., Coleman, J.S.: Photosynthetic thermotolerance is quantitatively and positively correlated with production of specific heat-shock proteins among nine genotypes of Lycopersicon (tomato).-Photosynthetica 38: 127-134, 2000.
Quietsch, C., Hong, S.W., Vierling, E., Lindquist, S.: Heat shock protein 101 plays a crucial role in thermotolerance in Arabidopsis.-Plant Cell 12: 479-492, 2000.
Rigola, D., Pe, M.E., Sari-Gorla, M.: A cDNA clone from hazelnut (Corylus avellana L.) encoding a low molecular weight heat shock protein expressed in the reproductive structures.-Sex. Plant Reprod. 11: 29-30, 1998.
Ristic, Z., Yang, G., Martin, B., Fullerton, S.: Evidence and association between specific heat shock proteins and the drought and heat tolerance phenotype in maize.-J. Plant Physiol. 153: 497-505, 1997.
Suzuki, T.C., Krawitz, D.C., Vierling, E.: The chloroplast small heat shock protein oligomer is not phosphorylated and does not dissociate during heat stress in vivo.-Plant Physiol. 116: 1151-1161, 1998.
Tsukaya, H., Takahashi, T., Naito, S., Komeda, Y.: Floral organ-specific and constitutive expression of an Arabidopsis thaliana heat shock Hsp 18.2=GUS fusion gene is retained even after homeotic conversion of flowers by mutation.-Mol. gen. Genet. 237: 26-32, 1993.
Vierling, E.: The roles of heat shock proteins in plants.-Annu. Rev. Plant Physiol. Plant mol. Biol. 42: 579-620, 1991.
Waters, E.: The molecular evolution of the small heat shock proteins in plants.-Genetics 141: 785-795, 1995.
Žárský, V., Garrido, D., Eller, N., Tupý, J., Vicente, O., Schoffl, F., Heberle-Bors, E.: The expression of small heat shock gene is activated during induction of tobacco pollen embryogenesis by starvation.-Plant Cell Environ. 18: 139-147, 1995.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ledesma, N., Kawabata, S. & Sugiyama, N. Effect of High Temperature on Protein Expression in Strawberry Plants. Biologia Plantarum 48, 73–79 (2004). https://doi.org/10.1023/B:BIOP.0000024278.62419.ee
Issue Date:
DOI: https://doi.org/10.1023/B:BIOP.0000024278.62419.ee