Abstract
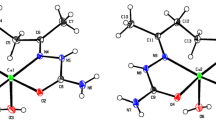
The crystal structures of several [Cu(RCN)4]X salts (R = Me, X = SO3CF3 −; R = Ph, X = BF4 −, ClO4 −, and PF6 −) were determined using single crystal X-ray diffraction. All of the compounds contain distorted tetrahedral Cu(I) centers and noncoordinating anions, with the acetonitrile and benzonitrile structures containing three and one unique CuL4 + complex in their respective asymmetric units. One important distortion is observed in the benzonitrile-Cu bonds, which are bent up to 23° away from linearity. The result is a flattened complex that maximizes the π–π tacking of the aromatic rings and is the dominant packing interactions between the complexes.
Similar content being viewed by others
References
Venkataraman, D.; Lee, S.; Moore, J.S.; Zhang, P.; Hirsh, K.A.; Gardner, G.B.; Covey, A.; Prentice, C.L. Chem. Mater. 1996, 8, 2030.
(a)Lopez, S.; Keller, S.W. Inorg. Chem. 1999, 38, 1883.(b)Lopez, S.; Keller, S.W. Cryst. Eng. 1999, 101.
Tabares, L.C.; Navarro, J.A.R.; Salas, J.M. J. Am. Chem. Soc. 2001, 123, 383.
Knaust, J.M.; Lopez, S.; Keller, S.W. Inorg. Chim. Acta 2001, 324, 81-89.
Jones, P.G.; Crespo, O. Acta Cryst. 1998, C54, 18-20.
Csöregh, I.; Kierkegaard, P.; Norrestam, R. Acta Cryst. 1975, B31, 314-317.
Black, J.R.; Levason, W.; Webster, M. Acta Cryst. 1995, C51, 623-625.
Chambers, R.D.; Gray, W.K.; Vaughan, J.F.S.; Korn, S.R.; Medebielle, M.; Batsanov, A.S.; Lehmann, C.W.; Howard, J.A.K. J. Chem. Soc., Perkin Trans. 1 1997, 2, 135.
Pohl, S; Lotz, R.; Saak, W.; Haase, D. Angew. Chem. Int. Ed. Engl. 1989, 28, 344.
Brownstein, S.; Han, N.F.; Gabe, E.; Page, Le Y. Can. J. Chem. 1989, 67, 2222.
Kubota, M.; Johnston, D.L. J. Inorg. Nucl. Chem. 1967, 28, 769.
Kubas, G. Inorg. Synth. 1979, 19.
Sheldrick, G.M. SHELXS-90, Program for Structure Solution; University of Gottingen: Germany, 1991.
Sheldrick, G.M. SHELXS-90, Program for Structure Solution; University of Gottingen: Germany, 1991, automatic Patterson method.
(a)Sheldrick, G.M. SHELXL-93, Program for Structure Refinement; University of Gottingen: Germany, 1994.(b)Barbour, L. X-SEED, Graphical Interface for SHEL-X; University of Missouri, Columbia, MO, 2000.
A further complication arose, in that one oxygen atom occupied virtually the same position as the carbon atom of the alternate position. Because of the similarity in scattering power of C and O (and the relatively equal occupancy) the site was refined as a fully occupied carbon atom.
All of the descriptions and pictures appearing in the manuscript will be of the ClO -4 structure. Analogous figures and crystallographic data for the BF -4 salt will be placed in the Supplementary Materials.
Hunter, C.A. Sanders, J.K.M. J. Am. Chem. Soc. 1990, 112, 5534.
Hunter, C.A.; Lawson, K.R.; Perkins, J.; Urch, C.J. J. Chem. Soc. Perkin Trans. 2001, 2, 651.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knaust, J.M., Knight, D.A. & Keller, S.W. Crystal and molecular structures of several tetrakis (nitrile)copper(I) complexes. Journal of Chemical Crystallography 33, 813–823 (2003). https://doi.org/10.1023/A:1027445410426
Issue Date:
DOI: https://doi.org/10.1023/A:1027445410426