Abstract
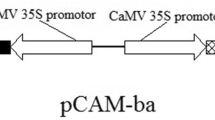
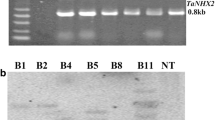
In the present work, the bacterial mannitol-1-phosphodehydrogenase(mtlD) gene was introduced into eggplant(Solanummelongena L.) by Agrobacteriumtumefaciens-mediated transformation. Several transformants weregenerated and the transgene integration was confirmed by PCR, dot blot andSouthern blot analysis. Transgenic lines of T0 and T1generations were examined for tolerance to NaCl-induced salt stress,polyethylene glycol-mediated drought and chilling stress under bothinvitro and in vivo growth conditions. Aconsiderable proportions of transgenic seeds germinated and seedlings grew wellon 200 mM salt-amended MS basal medium, whereas seeds ofuntransformed control plants failed to germinate. Further, leaf explants fromthe transgenics could grow and showed signs of shoot regeneration onsalt-amended MS regeneration medium, whereas wild type did not respond, and infact the explants showed necrosis and loss of chlorophyll after about one week.The transgenic leaves could also withstand desiccation, and transgenics couldgrow well under chilling stress, and hydroponic conditions with salt stress ascompared to wild type plants. Thus, the transgenic lines were found to betolerant against osmotic stress induced by salt, drought and chilling stress.The morphology of the transgenic plants was normal as controls, but thechlorophyll content was higher in some of the lines. These observations suggestthat mtlD gene can impart abiotic stress tolerance ineggplant.
Similar content being viewed by others
References
Aono M., Kubo A., Saji H., Tanaka K. and Kondo N. 1993. Enhanced tolerance to photoxidative stress of transgenic Nicotiana tobaccum with high chloroplastic reductase activity. Plant Cell Physiol. 34: 129-135.
Arnon D.I., McSwain B.D., Tsujimoto H.Y. and Wada K. 1974. Photochemical activity and components of membrane preparation from blue-green algae. I. Coexistence of two photosystems in relation to chlorophyll a and removal of phycocyanin. Biochem. Biophys. Acta 357: 231-245.
Bajaj S., Torgolli J., Liu L.-F., David Ho T.-H. and Wu R. 1999. Transgenic approaches to increase dehydration-stress tolerance in plants. Mol. Breed. 5: 493-503.
Bailey J.E. 1991. Toward a science of metabolic engineering. Science 252: 1668-1675.
Bohnert H.J. and Jensen R.G. 1996. Strategies for engineering water stress tolerance in plants. Trends Biotech. 14: 89-97.
Bohnert H.J. and Shen B. 1999. Transformation and compatible solutes. Scien. Hort. 78: 237-260.
Bray E.A. 1997. Plant responses to water deficit. Trends Plant Sci. 2: 48-54.
Capell J., Escobar C., Lui H., Burtin D., Lepri O. and Christou P. 1998. Over-expression of the oat arginine decarboxylase cDNA in transgenic rice (Oryza sativa L.) affects normal development patterns in vitro and results in putrescine accumulation in transgenic plants. Theor. Appl. Gene. 97: 246-254.
Collonnier C., Fock I., Kasyap V., Rotino G.L., Daunay M.C., Lian Y. et al. 2001. Applications of biotechnology in eggplant (Solanum melongena L.). Plant Cell Tiss. Org. Cul. 65: 97-107.
Doyle J.J. and Doyle J.L. 1990. Isolation of plant DNA from fresh tissue. Focus 12: 13-15.
Fillippone E. and Lurquin P.F. 1989. Stable transformation of eggplant (Solanum melongena L.) by cocultivation of tissues with Agrobacterium tumefaciens carrying a binary vector. Plant Cell Rep. 8: 370-373.
Fukushima E., Arata Y., Endo T., Sonnewald U. and Sato F. 2001. Improved salt tolerance of transgenic tobacco expressing apoplastic yeast-derived invertase. Plant Cell Physiol. 42: 245-2249.
Gisbert C., Rus A.M., Bolarin M.C., Lopez-Coronado J.M., Arrillaga I., Montesinos C. et al. 2000. The yeast HAL1 gene improves salt tolerance of transgenic tomato. Plant Physiol. 123: 393-402.
Grover A., Sahi C., Sanan N. and Grover A. 1999. Taming abiotic stresses in plants through genetic engineering: current strategies and perspective. Plant Sci. 143: 101-111.
Guri A. and Sink K.C. 1988. Agrobacterium transformation of eggplant. J. Plant Physiol. 133: 52-55.
Hiei Y., Komari T. and Kubo T. 1997. Transformation of rice mediated by Agrobacterium tumefaciens. Plant Mol. Biol. 35: 205-218.
Holmstrom K.O., Welin B., Mandal A., Kristiansdottir I., Teeri T.H., Trond L. et al. 1994. Production of the E.coli betaine aldehyde dehydrogenase., an enzyme required for the synthesis of the osmoprotectant glycine betaine in transgenic plants. Plant J. 6: 749-758.
Hoshda H., Tanaka Y., Hibino T., Hayashi Y., Tanaka A., Takabe T. et al. 2000. Enhanced tolerance to salt stress in transgenic rice that overexpresses chloroplast glutamine synthetase. Plant Mol. Biol. 43: 103-111.
Karakas B., Ozias-Akins P., Stushnoff C., Suefferheld M. and Rieger M. 1997. Salinity and drought tolerance of mannitol-accumulating transgenic tobacco. Plant Cell Environ. 20: 609-616.
Kavi Kishor P.B., Hong Z., Miao G., Hu C. and Verma D.P.S. 1995. Overexpression of Δ-1-pyrroline-5-carboxylate synthetase increases proline overproduction and confers osmotolerance in transgenic plants. Plant Physiol. 108: 1387-1394.
Kumria R., Waie B. and Rajam M.V. 2001. Plant regeneration from transformed embryogenic callus of an elite indica rice via Agrobacterium. Plant Cell. Tiss. Org. Cul. 67: 63-71.
McKersie B.D., Bowley S.R., Harjanto E. and Leprince O. 1996. Water-deficit tolerance and field performance of transgenic alfalfa over expressing superoxide dismutase. Plant Physiol. 111: 1177-1181.
Murashige T. and Skoog F. 1962. A revised medium for rapid growth and bioassays of tobacco tissue cultures. Physiol. Plant. 15: 473-497.
Murata N., Ishizaki-Nishizawa O., Higashi S., Hayashi H., Tasaka Y. and Nishida I. 1992. Genetically engineered alteration in the chilling sensitivity of plants. Nature 356: 710-713.
Pharr D.M., Prata R.T.N., Jennings D.B., Williamsom J.D. and Zamski E. 1999. Regulation of mannitol dehydrogenase: relationship to plant growth and stress tolerance. HortSci. 34: 1027-1032.
Pilon-Smits E.A.H., Ebskamp M.J.M., Paul M.J., Jeuken M.J.W., Weisbeek P.J. and Smeekens S.C.M. 1995. Improved performance of transgenic fructan-accumulating tobacco under drought stress. Plant Physiol. 107: 125-130.
Prasad K.V.S.K., Sharmila P., Kumar P.A. and Pardha Saradhi P. 2000. Transformation of Brassica juncea (L.) Czern with bacterial codA gene enhances its tolerance to salt stress. Mol. Breed. 6: 489-499.
Rajam M.V. 1997. Polyamines. In: Prasad M.N.V. (ed.), Plant Ecophysiology. John Wiley & Sons Inc., New York, pp. 343-374.
Rajam M.V. 2000. Over-expression of polyamine biosynthesis genes confers abiotic stress tolerance in transgenic plants. In: XXIV All India Cell Biology Conference. Jawaharlal Nehru University, New Delhi, p. 88.
Rajam M.V., Dagar S., Waie B., Yadav J.S., Kumar P.A., Shoeb F. et al. 1998. Genetic engineering of polyamine and carbohydrate metabolism for osmotic stress tolerance in higher plants. J. Biosci. 23: 473-482.
Romero C., Belles J.M., Vaya J.L., Serrano R. and Culianez-Macia F.A. 1997. Expression of the yeast trehalose-6-phosphate synthase gene in transgenic tobacco plants: pleotropic phenotypes include drought tolerance. Planta 201: 293-297.
Rotino G.L. and Gleddie S. 1990. Transformation of eggplant (Solanum melongena L.) using a binary Agrobacterium tumefaciens vector. Plant Cell Rep. 9: 26-29.
Sakamoto A. and Murata N. 2001. The use of bacterial choline oxidase, a glycinebetaine-synthesizing enzyme, to create stressresistant transgenic plants. Plant Physiol.: 180-188.
Sharma P. and Rajam M.V. 1995. Genotype, explant and position effects on organogenesis and somatic embryogenesis in eggplant (Solanum melongena L.). J. Exp. Bot. 46: 135-141.
Sambrook J., Fritsch E.F. and Maniatis T. 1989. Molecular Cloning: A Laboratory Mannual. Cold Spring Harbor laboratory, Cold Spring Harbor, New York, USA.
Shen B., Jensen R.G. and Bohnert H.J. 1997. Increased resistance to oxidative stress in transgenic plants by targeting mannitol biosynthesis to chloroplasts. Plant Physiol. 113: 1177-1183.
Sheveleva E., Chmara W., Bohnert H.J. and Jensen R.G. 1997. Increased salt and drought tolerance by D-ononitol production in transgenic Nicotiana tabacum. Plant Physiol. 115: 1211-1219.
Smart C.C. and Flores S. 1997. Overexpression of D-myo-inositol-3-phosphate synthase leads to elevated levels of inositol in Arabidopsis. Plant Mol. Biol. 33: 811-820.
Tarczynski M.C., Jensen R.G. and Bohnert H.J. 1992. Expression of a bacterial mtlD gene in transgenic tobacco leads to production and accumulation of mannitol. Proc. Natl. Acad. Sci., USA. 89: 2600-2604.
Tarczynski M.C., Jensen R.G. and Bohnert H.J. 1993. Stress protection of transgenic tobacco by production of the osmolyte mannitol. Science 259: 508-510.
Thomas J.C., Sepahi M., Arendall B. and Bohnert H.J. 1995. Enhancement of seed germination in high salinity by engineering mannitol expression in Arabidopsis thaliana. Plant Cell Environ. 18: 801-806.
Veena, Reddy V.S. and Sopory S.K. 1999. Glyoxylase I from Brassica juncea: molecular cloning, regulation and its overexpression confer tolerance in transgenic tobacco under stress. Plant J. 17: 385-395.
Vernon D.M., Tarczynski M.C., Jensen R.G. and Bohnert H.J. 1993. Cyclitol production in transgenic tobacco. Plant J. 4: 199-205.
Wallis J.G., Wang H. and Guerra D.J. 1997. Expression of a synthetic antifreeze protein in potato reduces electrolyte release at freezing temperatures. Plant Mol. Biol. 35: 323-330.
Xu D., Duan X., Wang B., Hong B., David Ho T.-H. and Wu R. 1996. Expression of a late embryogenesis abundant protein gene, HVA1 from barley confers tolerance to water deficit and salt stress in transgenic rice. Plant Physiol. 110: 249-257.
Zhu J.-K. 2001. Plant salt tolerance. Trends Plant Sci. 6: 66-71.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prabhavathi, V., Yadav, J., Kumar, P. et al. Abiotic stress tolerance in transgenic eggplant (Solanum melongena L.) by introduction of bacterial mannitol phosphodehydrogenase gene. Molecular Breeding 9, 137–147 (2002). https://doi.org/10.1023/A:1026765026493
Issue Date:
DOI: https://doi.org/10.1023/A:1026765026493