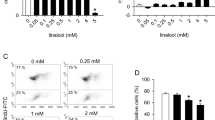
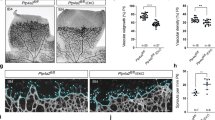
Abstract
Angiogenesis is a complex process that involves the activation of endothelial cells through the triggering of several intracellular signaling pathways including those involving tyrosine phosphorylation. In the present study, we analyzed the angiogenic properties of two phosphotyrosyl phosphatase (PTP) inhibitors that are composed of a peroxovanadium core containing different ancillary ligands. In cell monolayer and 3D culture systems examined in this study, the administration of potassium bisperoxo(1,10-phenanthroline)oxovanadate(V) [bpV(phen)] or potassium bisperoxo(pyridine-2-carboxylato)oxovanadate(V) [bpV(pic)], but not oxovanadiums, interfered markedly with endothelial cell growth, organization, and terminal differentiation. This effect was dependent upon both the compound's dose and the nature of the ancillary ligand. Rat aortic ring assay showed a significant inhibition by low dose of bpV(phen) on cell migration. In addition, a chick embryo angiogenesis assay demonstrated that bpV(phen) is a potent inhibitor of angiogenesis. Among PTP inhibitors, bpV(phen) had powerful angiostatic properties at a low concentration.
Similar content being viewed by others
References
Risau W. Mechanisms of angiogenesis. Nature 1997; 386: 671–4.
Folkman J, D'Amore PA. Blood vessel formation: What is its molecular basis? Cell 1996; 87: 1153–5.
Davis S, Aldrich TH, Jones PF et al. Isolation of angiopoietin-1, a ligand for the TIE2 receptor, by secretion-trap expression cloning. Cell 1996; 87: 1161–9.
Suri C, Jones PF, Patan S et al. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell 1996; 87: 1171–80.
Ullrich A, Schlessinger J. Signal transduction by receptors with tyrosine kinase activity. Cell 1990; 61: 203–12.
Mustonen T, Alitalo K. Endothelial receptor tyrosine kinases involved in angiogenesis. J Cell Biol 1995; 129: 895–8.
Jaye M, Schlessinger J, Dionne CA. Fibroblast growth factor receptor tyrosine kinases: Molecular analysis and signal transduction. Biochim Biophys Acta 1992; 1135: 185–99.
Partanen J, Armstrong E, Makela TP et al. A novel endothelial cell surface receptor tyrosine kinase with extracellular epidermal growth factor homology domains. Mol Cell Biol 1992; 12: 1698–707.
Dumont DJ, Yamaguchi TP, Conlon RA et al. Tek, a novel tyrosine kinase gene located on mouse chromosome 4, is expressed in endothelial cells and their presumptive precursors. Oncogene 1992; 7: 1471–80.
Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996; 86: 353–64.
Dumont DJ, Gradwohl G, Fong GH et al. Dominant-negative and targeted null mutations in the endothelial receptor tyrosine kinase, tek, reveal a critical role in vasculogenesis of the embryo. Genes Dev 1994; 8: 1897–909.
Fong GH, Rossant J, Gertsenstein M, Breitman ML. Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 1995; 376: 66–70.
Puri MC, Rossant J, Alitalo K et al. The receptor tyrosine kinase TIE is required for integrity and survival of vascular endothelial cells. Embo J 1995; 14: 5884–91.
Sato TN, Tozawa Y, Deutsch U et al. Distinct roles of the receptor tyrosine kinases Tie-1 and Tie-2 in blood vessel formation. Nature 1995; 376: 70–4.
Shalaby F, Rossant J, Yamaguchi TP et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 1995; 376: 62–6.
Carmeliet P, Ferreira V, Breier G et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 1996; 380: 435–9.
Relf M, LeJeune S, Scott PA et al. Expression of the angiogenic factors vascular endothelial cell growth factor, acidic and basic fibroblast growth factor, tumor growth factor beta-1, platelet-derived endothelial cell growth factor, placenta growth factor, and pleiotrophin in human primary breast cancer and its relation to angiogenesis. Cancer Res 1997; 57: 963–9.
Hubbard SR, Mohammadi M, Schlessinger J. Autoregulatory mechanisms in protein-tyrosine kinases. J Biol Chem 1998; 273: 11987–90.
Mohammadi M, Froum S, Hamby JM et al. Crystal structure of an angiogenesis inhibitor bound to the FGF receptor tyrosine kinase domain. Embo J 1998; 17: 5896–904.
Jove R, Hanafusa H. Cell transformation by the viral src oncogene. Annu Rev Cell Biol 1987; 3: 31–56.
Hunter T. Protein modification: Phosphorylation on tyrosine residues. Curr Opin Cell Biol 1989; 1: 1168–81.
Fischer EH, Charbonneau H, Tonks NK. Protein tyrosine phosphatases: A diverse family of intracellular and transmembrane enzymes. Science 1991; 253: 401–6.
Tonks NK, Diltz CD, Fischer EH. Characterization of the major protein-tyrosine-phosphatases of human placenta. J Biol Chem 1988; 263: 6731–7.
Guan KL, Dixon JE. Evidence for protein-tyrosine-phosphatase catalysis proceeding via a cysteine-phosphate intermediate. J Biol Chem 1991; 266: 17026–30.
Denu JM, Dixon JE. A catalytic mechanism for the dual-specific phosphatases. Proc Natl Acad Sci USA 1995; 92: 5910–4.
Zhang ZY, Wang Y, Dixon JE. Dissecting the catalytic mechanism of protein-tyrosine phosphatases. Proc Natl Acad Sci USA 1994; 91: 1624–7.
Tonks NK, Diltz CD, Fischer EH. Purification of the major protein-tyrosine-phosphatases of human placenta. J Biol Chem 1988; 263: 6722–30.
Barford D, Flint AJ, Tonks NK. Crystal structure of human protein tyrosine phosphatase 1B. Science 1994; 263: 1397–404.
Shaver A, Ng JB, Hall DA, Posner BI. The chemistry of peroxovanadium compounds relevant to insulin mimesis. Mol Cell Biochem 1995; 153: 5–15.
Posner BI, Faure R, Burgess JW et al. Peroxovanadium compounds. A new class of potent phosphotyrosine phosphatase inhibitors which are insulin mimetics. J Biol Chem 1994; 269: 4596–604.
Faure R, Vincent M, Dufour M et al. Arrest at the G2/M transition of the cell cycle by protein-tyrosine phosphatase inhibition: Studies on a neuronal and a glial cell line. J Cell Biochem 1995; 59: 389–401.
Olivier M, Romero-Gallo BJ, Matte C et al. Modulation of interferon-gamma-induced macrophage activation by phosphotyrosine phosphatases inhibition. Effect on murine Leishmaniasis progression. J Biol Chem 1998; 273: 13944–9.
Jaffe EA, Nachman RL, Becker CG, Minick CR. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest 1973; 52: 2745–56.
Janvier R, Sourla A, Koutsilieris M, Doillon CJ. Stromal fibroblasts are required for PC-3 human prostate cancer cells to produce capillary-like formation of endothelial cells in a three-dimensional co-culture system. Anticancer Res 1997; 17: 1551–7.
Nicosia RF, Ottineti A. Growth of microvessels in serum-free-matrix culture of aorta. A quantitative assay of angiogenesis in vitro. Lab Invest 1990, 63: 115–22.
Draetta G, Eckstein J. Cdc25 protein phosphatases in cell proliferation. Biochim Biophys Acta 1997; 1332: M53-M63.
Fotsis T, Pepper M, Adlercreutz H et al. Genistein, a dietary ingested isoflavonoid, inhibits cell proliferation and in vitro angiogenesis. J Nutr 1995; 125: 790S-7S.
Montesano R, Pepper MS, Belin D et al. Induction of angiogenesis in vitro by vanadate, an inhibitor of phosphotyrosine phosphatases. J Cell Physiol 1988; 134: 460–6.
Jackson JK, Min W, Cruz TF et al. A polymer-based drug delivery system for the antineoplastic agent bis(maltolato)oxovanadium in mice. Br J Cancer 1997; 75: 1014–20.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doillon, C.J., Faure, R.L., Posner, B.I. et al. Peroxovanadium compounds as inhibitors of angiogenesis. Angiogenesis 3, 361–369 (1999). https://doi.org/10.1023/A:1026577418765
Issue Date:
DOI: https://doi.org/10.1023/A:1026577418765