Abstract
The inappropriate expression of growth factors, or activating mutations of their receptors, have been implicated as causative factors in mouse and human mammary cancer. For example, it has been known for some time that three members of the fibroblast growth factor (FGF)3 family behave like oncogenes in virally induced mammary cancer of mice. In normal circumstances, signaling via FGF receptors is known to mediate growth, differentiation, and patterning, during embryogenesis and fetal development. A powerful approach to dissecting the roles for these signaling pathways is to determine the developmental consequences of abrogating their function in transgenic mice. In this review, we describe the use of dominant negative FGF receptors to evaluate the contribution of specific FGF signals in normal mammary gland development. These studies have revealed that normal lobuloalveolar development requires FGF signaling to the mammary epithelium, a function that is presumably usurped by MMTV in mouse mammary tumorigenesis.
Similar content being viewed by others
REFERENCES
D. Medina, (1996). The mammary gland: A unique organ for the study of development and tumorigenesis. J. Mam. Gland Biol. Neoplasia 1:5–19.
G. Cunha and Y. Hom (1996). Role of mesenchymal-epithelial interactions in mammary gland development. J. Mam. Gland Biol. Neoplasia 1:21–35.
C. Daniel and G. Silberstein (1987). Postnatal development of the rodent mammary gland., In M. Neville and C. Daniel (eds.). The Mammary Gland: Regulation, and Function., Plenum Press, New York, pp. 3–36.
W. Imagawa J. Yang, R. Guzman, and S. Nandi (1994). Control of mammary gland development, In E. Knobil and J. Neill (eds.), The Physiology of Reproduction, Raven Press, New York, pp. 1033–1065.
N. Hynes (1996). ErbB2 activation and signal transduction in normal and malignant mammary cells. J. Mam. Biol. Neoplasia 1:199–206.
E. Peles and Y. Yarden (1993). Neu and its ligands: from an oncogene to neural factors. Bioessays 15:815–824.
C. A. MacArthur, D. B. Shankar, and G. M. Shackleford (1995). FGF-8, activated by proviral insertion, cooperates with the wnt-1 transgene in murine mammary tumorigenesis. J. Virol. 69:2501–2507.
C. Dickson, R. Smith, S. Brookes, and G. Peters (1984). Tumorigenesis by mouse mammary tumor virus: proviral activation of a cellular gene in the common integration region int-2. Cell 37:529–536.
D. Gallahan, C. Kozak, and R. Callahan (1987). A new common integration region (int-3) for mouse mammary tumor virus on mouse chromosome 17. J. Virol. 61:218–220.
H. Roelink, E. Wagenaar, S. Lopes da Silva, and R. Nusse (1990). Wnt-3, a gene activated by proviral insertion in mouse mammary tumors, is homologous to int-1/Wnt-1 and is normally expressed in mouse embryos and adult brain. Proc. Natl. Acad. Sci. U.S.A. 87:4519–4523.
R. Nusse, A. van Ooyen, D. Cox, Y. K. T. Fung, and H. Varmus (1984). Mode of proviral activation of a putative mammary oncogene (int-1) on mouse chromosome 15. Nature 307:131–136.
G. Peters, S. Brookes R. Smith, M. Placzek, and C. Dickson (1989). The mouse homolog of the hst/k-FGF gene is adjacent to int-2 and activated by proviral insertion in some virally induced mammary tumors. Proc. Natl. Acad. Sci. U.S.A. 86:5678–5682.
G. Peters (1991). Inappropriate expression of growth factor genes in tumors induced by mouse mammary tumor virus. Sem. Virol. 2:319–328.
T. P. Yamaguchi and J. Rossant (1995). Fibroblast growth-factors in mammalian development. Curr. Opin. Genet. Devel. 5:485–491.
R. Nusse and H. E. Varmus (1992). Wnt genes. Cell 69:1073–1087.
B. Gavin and A. McMahon (1992). Differential regulation of the Wnt gene family during pregnancy and lactation suggests a role in postnatal development of the mammary gland. Mol. Cell. Biol. 12:2418–2423.
L. Bouchard, L. Lamarre, P. Tremblay, and P. Jolicoeur (1989). Stochastic appearance of mammary tumors in transgenic mice carrying the MMTV/c-neu oncogene. Cell 57:931–936.
W. Muller, E. Sinn, P. Pattengale, R. Wallace, and P. Leder (1988). Single step induction of mammary adenocarcinoma in transgenic mice bearing the activated c-neu oncogene. Cell 54:105–115.
A. S. Tsukamoto, R. Grosschedl, R. C. Guzman, T. Parslow, and H. E. Varmus (1988). Expression of the int-1 gene in transgenic mice is associated with mammary gland hyperplasia and adenocarcinomas in male and female mice. Cell 55:619–625.
W. J. Muller, F. S. Lee, C. Dickson, G. Peters, P. Pattengale, and P. Leder (1990). The int-2 gene product acts as an epithelial growth factor in transgenic mice. EMBO J. 9:907–913.
D. M. Ornitz, R. D. Cardiff, A. Kuo, and P. Leder (1992). Int-2, an autocrine and/or ultra-short-range effector in transgenic mammary tissue transplants. J. Natl. Cancer Inst. 84:887–892.
G. Stamp, V. Fantl, R. Poulsom, S. Jamieson, R. Smith, G. Peters, and C. Dickson (1992). Nonuniform expression of a mouse mammary tumor virus-driven int-2/Fgf-3 transgene in pregnancy-responsive breast tumors. Cell Growth Diff. 3:929–938.
D. Kitsberg, and P. Leder (1996). Keratinocyte growth-factor induces mammary and prostatic hyperplasia and mammary adenocarcinoma in transgenic mice. Oncogene 13:2507–2515.
G. Silberstein and C. Daniel (1987). Regulation of mammary growth and function by transforming growth factor-beta. Science 32:145–151.
Y. Yanmin, E. Spitzer, D. Meyer, M. Sachs, C. Niemann, G. Hartmann, K. Weidner, C. Birchmeier, and W. Birchmeier (1995). Sequential requirement of hepatocyte growth factor and neuregulin in the morphogenesis and differentiation of the mammary gland. J. Cell. Biol. 131:215–226.
C. Basilico, D. Moscatelli (1992). The FGF family of growth-factors and oncogenes. Adv. Cancer Res. 59:115–165.
W. H. Burgess and T. Maciag (1989). The heparin binding (fibroblast) growth factor family proteins. Ann. Rev. Biochem. 58:575–606.
I. Thesleff, A. Vaahtokari, and A. M. Partanen (1995). Regulation of organogenesis. Common molecular mechanisms regulating the development of teeth and other organs. Int. J. Devel. Biol. 39:35–50.
A. O. M. Wilkie, G. M. Morrisskay, E. Y. Jones, and J. K. Heath (1995). Functions of fibroblast growth-factors and their receptors. Curr. Biol. 5:500–507.
M. Muenke, and U. Schell (1995). Fibroblast-growth-factor receptor mutations in human skeletal disorders. Trends Genet. 11:308–313.
M. Jaye, J. Schlessinger, and C. Dionne (1992). Fibroblast growth factor receptor tyrosine kinases: molecular analysis and signal transduction. Biochem. Biophys. Acta 1135:185–199.
D. Johnson and L. Williams (1993). Structural and functional diversity in the FGF receptor multigene family. Adv. Cancer Res. 60:1–41.
D. M. Ornitz, and P. Leder (1992). Ligand specificity and heparin dependence of fibroblast growth-factor receptor-1 and receptor-3. J. Biol. Chem. 267:16305–16311.
M. Klagsbrun and A. Baird, (1991). A dual receptor system is required for basic fibroblast growth factor activity. Cell 67:229–231.
S. Coleman-Krnacik, and J. M. Rosen (1994). Differential temporal and spatial gene-expression of fibroblast growth-factor family members during mouse mammary-gland development. Mol. Endocrinol. 8:218–229.
M. Mathieu, E. Chatelain, D. Ornitz, J. Bresnick, I. Mason, P. Kiefer, and C. Dickson (1995). Receptor-binding and mitogenic properties of mouse fibroblast-growth-factor-3—modulation of response by heparin. J. Biol. Chem. 270:24197–24203.
S. L. Mansour, J. M. Goddard, and M. R. Capecchi (1993). Mice homozygous for a targeted disruption of the proto-oncogene int-2 have developmental defects in the tail and inner ear. Development 117:13–28.
L. Guo, L. Degenstein, and E. Fuchs (1996). Keratinocyte growth factor is required for hair development but not for wound healing. Genes Devel. 10:165–175.
J. M. Hebert, T. Rosenquist, J. Gotz, and G. R. Martin (1994). FGF5 as a regulator of the hair-growth cycle—evidence from targeted and spontaneous mutations. Cell 78:1017–1025.
C. Deng, B. A. Wynshaw, F. Zhou, A. Kuo, and P. Leder (1996). Fibroblast growth factor receptor 3 is a negative regulator of bone growth. Cell 84:911–921.
J. S. Colvin, B. A. Bohne, G. W. Harding, D. G. McEwen, and D. M. Ornitz (1996). Skeletal overgrowth and deafness in mice lacking fibroblast growth factor receptor 3. Nature Genet. 12:390–397.
B. Feldman, W. Poueymirou, V. E. Papaioannou, T. M. Dechiara, and M. Goldfarb (1995). Requirement of FGF-4 for postimplantation mouse development. Science 267:246–249.
T. P. Yamaguchi, K. Harpal, M. Henkemeyer, and J. Rossant (1994). FGFR-1 is required for embryonic growth and mesodermal patterning during mouse gastrulation. Genes Devel. 8:3032–3044.
C. X. Deng, A. Wynshawboris, M. M. Shen C. Daugherity, D. M. Ornitz, and P. Leder (1994). Murine FGFR-1 is required for early postimplantation growth and axial organization. Genes Devel. 8:3045–3057.
S. Werner, W. Weinberg, X. Liao, K. Peters, M. Blessing, S. Yuspa, R. Weiner, and L. Williams (1993). Targeted expression of a dominant-negative FGF receptor mutant in the epidermis of transgenic mice reveals a role of FGF in keratinocyte organization and differentiation. EMBO J. 12:2635–2643.
S. Werner, H. Smola, X. Liao, M. Longaker, T. Krieg, P. Hofschneider, and L. Williams (1994). The function of KGF in morphogenesis of epithelium and reepithelialization of wounds. Science 266:819–822.
K. Peters, S. Werner, X. Liao, S. Wert, J. Whitsett, and L. Williams (1994). Targeted expression of a dominant negative FGF receptor blocks branching morphogenesis and epithelial differentiation of the mouse lung. EMBO J. 13:3296–3301.
O. Kashles, Y. Yarden, A. Ullrich, and J. Schlessinger (1991). A dominant negative mutation suppresses the function of normal epidermal growth factor receptors by heterodimerization. Mol. Cell. Biol. 11:1454–1453.
H. Ueno, M. Gunn, K. Dell, A. Tseng, and L. Williams (1992). A truncated form of fibroblast growth-factor receptor-1 inhibits signal transduction by multiple types of fibroblast growth-factor receptor. J. Biol. Chem. 267:1470–1476.
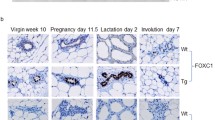
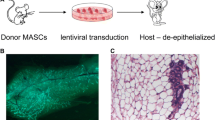
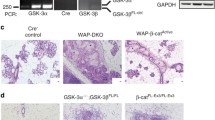
D. Jackson, J. Bresnick, I. Rosewell, T. Crafton, R. Poulson, G. Stamp, and C. Dickson (1997). Fibroblast growth factor signaling has a role in lobuloalveolar development of the mammary gland. J. Cell Sci. 110:1261–1268.
T. Ulich, E. Yi, R. Cardiff, S. Yin, N. Bikhazi, R. Biltz, C. Morris, and G. Pierce (1994). Keratinocyte growth factor is a growth factor for mammary epithelium in vivo. Am. J. Pathol. 144:862–868.
I. Mason, F. Fuller-Pace, R. Smith, and C. Dickson (1994). FGF-7 (keratinocyte growth factor) expression during mouse development suggests roles in myogenesis, forebrain regionalisation and epithelial interactions. Mech. Devel. 45:15–30.
M. Yamasaki, A. Miyake, S. Tagashira, and N. Itoh (1996). Structure and expression of the rat mRNA encoding a novel member of the fibroblast growth factor family. J. Biol. Chem. 271:15918–15921.
D. Ornitz, J. Xu, J. Colvin, D. McEwen, C. MacArthur, F. Coulier, G. Gao, and M. Golfarb (1996). Receptor specificity of the fibroblast growth-factor family. J. Biol. Chem. 271:15292–15297.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jackson, D., Bresnick, J. & Dickson, C. A Role for Fibroblast Growth Factor Signaling in the Lobuloalveolar Development of the Mammary Gland. J Mammary Gland Biol Neoplasia 2, 385–392 (1997). https://doi.org/10.1023/A:1026351414004
Issue Date:
DOI: https://doi.org/10.1023/A:1026351414004