Abstract
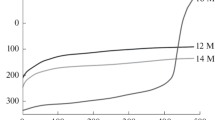
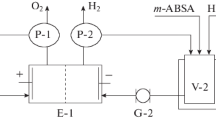
A procedure was proposed for electrochemical reduction of 2,5-dichloronitrobenzene to 2,5-dichloroaniline in aqueous-ethanolic solution of sulfuric acid. The procedure for preparing 2,5-dichloroanilinium sulfate was optimized. The influence exerted by the cathode material, nature and amount of the organic solvent, sulfuric acid concentration, and electrolysis temperature on the yield and quality of the target product was discussed.
Similar content being viewed by others
References
Tomilov, A.P., Fioshin, M.Ya., and Smirnov, V.Ya., Elektrokhimicheskii sintez organicheskikh veshchestv (Electrochemical Synthesis of Organic Substances), Leningrad: Khimiya, 1976.
Izgaryshev, N.A. and Fioshin, M.Ya., Dokl. Akad. Nauk SSSR, 1953, vol. 90, pp. 189-190.
Technique of Organic Chemistry, vol. 2: Komarewsky, V.I., Riesz, C.H., and Moritz, F.L., Catalytic Reactions; Masson, C.R., Boekelheide, V., and Noyes, W.A., Photochemical Reactions; Swann, Sh., Electrolytic Reactions, New York: Interscience, 1956.
Gul'tyai, V.P. and Leibzon, V.N., Elektrokhimiya, 1996, vol. 32, pp. 65-74.
Tomilov, A.P. and Smirnov, Yu.D., Elektrokhimiya, 1997, vol. 33, pp. 1260-1261.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Islamgulova, V.R., Tomilov, A.P., Akhmerova, S.G. et al. Electrochemical Synthesis of 2,5-Dichloroaniline. Russian Journal of Applied Chemistry 76, 1076–1078 (2003). https://doi.org/10.1023/A:1026350110061
Issue Date:
DOI: https://doi.org/10.1023/A:1026350110061