Abstract
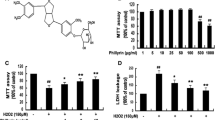
Phenelzine, deprenyl, and antioxidants (SOD, catalase, ascorbate, or rutin) reduced the loss of cell viability in differentiated PC12 cells treated with 250 μM MPP+, whereas N-acetylcysteine and dithiothreitol did not inhibit cell death. Phenelzine reduced the condensation and fragmentation of nuclei caused by MPP+ in PC12 cells. Phenelzine and deprenyl prevented the MPP+-induced decrease in mitochondrial membrane potential, cytochrome c release, formation of reactive oxygen species, and depletion of GSH in PC12 cells. Phenelzine revealed a scavenging action on hydrogen peroxide and reduced the hydrogen peroxide–induced cell death in PC12 cells, whereas deprenyl did not depress the cytotoxic effect of hydrogen peroxide. Both compounds reduced the iron and EDTA-mediated degradation of 2-deoxy-d-ribose degradation. The results suggest that phenelzine attenuates the MPP+-induced viability loss in PC12 cells by reducing the alteration of mitochondrial membrane permeability that seems to be mediated by oxidative stress.
Similar content being viewed by others
REFERENCES
Bernardi, P. 1996. The permeability transition pore: Control points of a cyclosporin A-sensitive mitochondrial channel involved in cell death. Biochim. Biophys. Acta 1275:5-9.
Lotharius, J., Dugan, L. L., and O'Malley, K. L. 1999. Distinct mechanisms underlie neurotoxin-mediated cell death in cultured dopaminergic neurons. J. Neurosci. 19:1284-1293.
Atlante, A., Calissano, P., Bobba, A., Giannattasio, S., Marra, E., and Passarella, S. 2001. Glutamate neurotoxicity, oxidative stress and mitochondria. FEBS Lett. 497:1-5.
Cassarino, D. S., Parks, J. K., Parker, W. D. Jr., and Bennett, J. P. Jr., 1999. The Parkinsonian neurotoxin MPP+ opens the mitochondrial permeability transition pore and releases cytochrome c in isolated mitochondria via an oxidative mechanism. Biochim. Biophys. Acta 1453:49-62.
Chakraborati, T., Das, S., Mondal, M., Roychoudhury, S., and Chakraborti, S. 1999. Oxidant, mitochondria and calcium: An overview. Cell. Signal. 11:77-85.
Chandra, J., Samali, A., and Orrenius, S. 2000. Triggering and modulation of apoptosis by oxidative stress. Free Radic. Biol. Med. 29:323-333.
Lee, C. S., Han, J. H., Jang, Y. Y., Song, J. H., and Han, E. S. 2002. Differential effect of catecholamines and MPP+ on membrane permeability in brain mitochondria and cell viability in PC12 cells. Neurochem. Int. 40:361-369.
Birkmayer, W., Knoll, J., Riederer, P., Youdim, M. B., Hars, V., and Marton, J. 1985. Increased life expectancy resulting from addition of L-deprenyl to Madopar treatment in Parkinson's disease: A longterm study. J. Neural. Transm. 64:113-127.
Jacobsson, S. O. and Fowler, C. J. 1999. Dopamine and glutamate neurotoxicity in cultured chick telencephali cells: Effects of NMDA antagonists, antioxidants and MAO inhibitors. Neurochem. Int. 34:49-62.
Lai, C.-T. and Yu, P. H. 1997. Dopamine-and L-β-3,4-dihydroxyphenylalanine hydrochloride (L-DOPA)-induced cytotoxicity towards catecholaminergic neuroblastoma SH-SY5Y cells: Effects of oxidative stress and antioxidative factors. Biochem. Pharmacol. 53:363-372.
Maher, P. and Davis, J. B. 1996. The role of monoamine metabolism in oxidative glutamate toxicity. Neuroscience 16: 6394-6401.
Le, W., Jankovic, J., Xie, W., Kong, R., and Appel, S. H. 1997. (-)-Deprenyl protection of 1-methyl-4 phenylpyridium ion (MPP+)-induced apoptosis independent of MAO-B inhibition. Neurosci. Lett. 224:197-200.
Wu, R. M., Chen, R. C., and Chiueh, C. C. 2000. Effect of MAO-B inhibitors on MPP+ toxicity in vivo. Ann. NY Acad. Sci. 899:255-261.
Matsubara, K., Senda, T., Uezono, T., Awaya, T., Ogawa, S., Chiba, K., Shimizu, K., Hayase, N., and Kimura, K. 2001. L-Deprenyl prevents the cell hypoxia induced by dopaminergic neurotoxins, MPP(+) and beta-carbolinium: A microdialysis study in rats. Neurosci. Lett. 302:65-68.
Lee, C. S., Han, E. S., Jang, Y. Y., Han, J. H., Ha, H. W., and Kim, D. E. 2000. Protective effect of harmalol and harmaline on MPTP neurotoxicity in the mouse and dopamine-induced damage of brain mitochondria and PC12 Cells. J. Neurochem. 75: 521-531.
Kim, D. H., Jang, Y. Y., Han, E. S., and Lee, C. S. 2001. Protective effect of harmaline and harmalol against dopamine-and 6-hydroxydopamine-induced oxidative damage of brain mitochondria and synaptosomes, and viability loss of PC12 cells. Eur. J. Neurosci. 13:1861-1872.
Davis, J. M., Janicak, P. G., and Bruninga, K. 1987. The efficacy of MAO inhibitors in depression: A meta-analysis. Psychiatric Ann. 17:825-831.
Liebowitz, M. R. 1993. Depression with anxiety and atypical depression. J. Clin. Psychiatry 54(Suppl.):10-14.
Fredriksson, A., Palomo, T., and Archer, T. 2000. Effects of MAO inhibitors upon MPTP mice chronically treated with suprathreshold doses of L-dopa. Behav. Pharmacol. 11: 571-581.
Tatton, W. G., Chalmers-Redman, R. M. E., Ju, W. J. H., Mammen, M., Carlile, G. W., Pong, A. W., and Tatton, N. A. 2002. Propargylamines induce antiapoptotic new protein synthesis in serum-and nerve growth factor (NGF)-withdrawn, NGF-differentiated PC-12 cells. J. Pharmacol. Exp. Ther. 301: 753-764.
Mosmann, T. 1983. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 65:55-63.
Oberhammer, F. A., Pavelka, M., Sharma, S., Tiefenbacher, R., Purchio, A. F., Bursch, W., and Schulte-Hermann, R. 1992. Induction of apoptosis in cultured hepatocytes and in regressing liver by transforming growth factor β1. Proc. Natl. Acad. Sci. USA 89:5408-5412.
Fu, W., Luo, H., Parthasarathy, S., and Mattson, M. P. 1998. Catecholamines potentiate amyloid β-peptide neurotoxicity: Involvement of oxidative stress, mitochondrial dysfunction, and perturbed calcium homeostasis. Neurobiol. Dis. 5:229-243.
van Klaveren, R. J., Hoet, P. H., Pype, J. L., Demedts, M., and Nemery, B. 1997. Increase in gamma-glutamyltransferase by glutathione depletion in rat type II pneumocytes. Free Radic. Biol. Med. 22:525-534.
Aruoma, O. I. 1994. Deoxyribose assay for detecting hydroxyl radicals. Vol. 233, Pages 57-66, in Packer, L. (ed.), Methods enzymol. Academic Press, San Diego.
Du, Y., Dodel, R. C., Bales, K. R., Jemmerson, R., Hamiton-Byrd, E., and Paul, S. M. 1997. Involvement of a caspase-3-like cysteine protease in 1-methyl-4-phenylpyridinium-mediated apoptosis of cultured cerebellar granule neurons. J. Neurochem. 69:1382-1388.
Zeevalk, G. D., Bernard, L. P., Albers, D. S., Mirochnitchenko, O., Nicklas, W. J., and Sonsalla, P. K. 1997. Energy stress-induced dopamine loss in glutathione peroxidase-overexpressing transgenic mice and in glutathione-depleted mesencephalic cultures. J. Neurochem. 68:426-429.
Pereira, C. F. and Oliveira, C. R. 2000. Oxidative glutamate toxicity involves mitochondrial dysfunction and perturbation of intracellular Ca2+ homeostasis. Neurosci. Res. 37:227-236.
Halliwell, B. and Gutteridge, J. M. C. 1999. Detection of free radicals and other reactive species: Trapping and fingerprinting. Pages 351-429, in Halliwell, B. and Gutteridge, J. M. C. (eds.), Free radicals in biology and medicine. 3rd ed. Oxford University Press, New York.
Kadota, T., Yamaai, T., Saito, Y., Akita, Y., Kawashima, S., Moroi, K., Inagaki, N., and Kadota, K. 1996. Expression of dopamine transporter at the tips of growing neurites of PC12 cells. J. Histochem. Cytochem. 44:989-996.
Amarante-Mendes, G. P., Kim, C. N., Liu, L., Huang, Y., Perkins, C. L., Green, D. R., and Bhalla, K. 1998. Bcr-Abl exerts its antiapoptotic effect against diverse apoptotic stimuli through blockade of mitochondrial release of cytochrome c and activation of caspase-3. Blood 91:1700-1705.
Tan, S., Sagara, Y., Liu, Y., Maher, P., and Schubert, D. 1998. The regulation of reactive oxygen species production during programmed cell death. J. Cell Biol. 141:1423-1432.
Jurma, O. P., Hom, D. G., and Andersen, J. K. 1997. Decreased glutathione results in calcium-mediated cell death in PC12. Free Radic. Biol. Med. 23:1055-1066.
Fonck, C. and Baudry, M. 2001. Toxic effects of MPP+ and MPTP in PC12 cells independent of reactive oxygen species formation. Brain Res. 905:199-206.
Obata, T., Yamanaka, Y., Kinemuchi, H., and Oreland, L. 2001. Release of dopamine by perfusion with 1-methyl-4-phenylpyridinium ion (MPP+) into the striatum is associated with hydroxyl free radical generation. Brain Res. 906:170-175.
Lotharius, J. and O'Malley, K. L. 2000. The Parkinsonism-inducing drug 1-methyl-4-phenylpyridinium triggers intracellular dopamine oxidation: A novel mechanism of toxicity. J. Biol. Chem. 275: 38581-38588.
Obata, T. 2002. Dopamine efflux by MPTP and hydroxyl radical generation. J. Neural Transm. 109:1159-1180.
Tatton, W. G. and Chalmers-Redman, R. M. E. 1996. Modulation of gene expression rather than monoamine oxidase inhibition. Neurology 47(Suppl. 3):S171-S183.
Olanow, C. W. and Tatton, W. G. 1999. Etiology and pathogenesis of Parkinson's disease. Annu. Rev. Neurosci. 22:123-144.
Banaclocha, M. M., Hernandez, A. I., Martinez, N., and Ferrandiz, M. L. 1997. N-acetylcysteine protects against age-related increase in oxidized proteins in mouse synaptic mitochondria. Brain Res. 762:256-258.
Seaton, T. A., Cooper, J. M., and Schapira, A. H. 1997. Free radical scavengers protect dopaminergic cell lines from apoptosis induced by complex I inhibitors. Brain Res. 777:110-118.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, C.S., Han, E.S. & Lee, W.B. Antioxidant Effect of Phenelzine on MPP+-Induced Cell Viability Loss in Differentiated PC12 Cells. Neurochem Res 28, 1833–1841 (2003). https://doi.org/10.1023/A:1026119708124
Issue Date:
DOI: https://doi.org/10.1023/A:1026119708124