Abstract
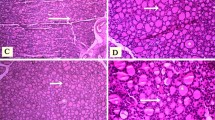
The aim of this study was to investigate the effects of centrally applied somatostatin-28 on morphometric characteristics of the thymus, the thymocyte subpopulations, as well as, on apoptosis and phases of cell cycle in thymocytes. For this purpose, peripubertal male rats were cannulated intracerebroventriculary and treated with repeated, nanomolar concentrations of somatostatin-28 (experimental group) or saline (control group). Animals were sacrificed and their thymuses were used for the analysis of thymocyte subpopulations, cell cycle and apoptosis by flow cytometry and for the evaluation of morphometric parameters by stereological analysis. Our results showed that somatostatin-28 caused decrease of the thymic mass and volume, as well as total thymocytes number. Stereological analysis revealed volume decrease of thymic cortex and medulla accompanied with cellularity decrease. Somatostatin in the deeper cortex decreased the number of thymocytes, per volume unit, while in outer cortex raised their number. A significant increase in the percentage of double-negative and both single-positive thymocyte subpopulations, in parallel with a diminished percentage of double-positive cells was found. The cellularity of double-positive and single-positive thymocyte subpopulations was decreased. Somatostatin-28 treatment augmented the percentage of apoptotic cells, while the percentage of the cells represented in phases of cell cycle was reduced.
These results suggest that somatostatin-28 induce thymus hypotrophy as result of decreasing cortex and medulla volume and cellularity. Changes in the percentage and cellularity of thymocyte subpopulations and numerical density of thymocytes in outer and deeper cortex, indicate that somatostatin-28 evoked disturbance in transition of double-negative to double-positive thymocytes.
Similar content being viewed by others
References
Cheng NW, Boyages SC (1995) Somatostatin-14 and its analog octreotide exert a cytostatic effect on GH3 rat pituitary tumor cell proliferation via a transient G0/G1 cell cycle block. Endocrinology 136: 4174–4181.
Duquesnon RJ (1972) Immunodeficiency of the thymus dependent system of the Ames dwarf mouse. J Immunol 108: 1578–1590.
Farr AG, Rudensky A (1998) Medullary thymic epithelium: A mosaic of epithelial "self’? J Exp Med 188: 1–4.
Ferone D, Van Hagen PM, Van Koetsveld PM, Zuijderwijk J, Mooy DM, Lichtenauer-Kaligis EG, Colao A, Bogers AJ, Lombardi G, Lamberts SW, Hofland LJ (1999) In vitro characterization of somatostatin receptors in the human thymus and effects of somatostatin and octreotide on cultured thymic epithelial cells. Endocrinology 140: 373–380.
Ferone D, Pivonello R, Van Hagen PM, Waaijers M, Zuijderwijk J, Colao A, Lombardi G, Bogers AJ, Lamberts SW, Hofland LJ (2000) Age-related decrease of somatostatin receptor number in the normal human thymus. Am J Physiol Endocrinol Metab 279: E791–E798.
Frohman LA, Downs TA, Chomcynski P (1992) Regulation of growth hormone secretion. Front Neuroendocrinol 13: 344–405.
Gagnerault MC, Postel-Vinay MC, Dardenne M (1996) Expression of growth hormone receptors in murine lymphoid cells analyzed by flow cytofluorometry. Endocrinology 137: 1719–1726.
Goverman J, Brabb T, Huseby ES, Farr AG (1997) TCR signaling regulates thymic organization: lessons from TCR-transgenic mice. Immunol Today 18: 204–208.
Hirokawa K, Makinodan T (1975) Thymus involution: Effect on T cell differentiation. J Immunol 6: 1659–1664.
Jameson SC, Bevan MJ (1998) T-cell selection. Curr Opin Immunol 10: 214–219.
Janković BD, Isaković K, Mićić M, Knežević Z (1980) The embryonic lympho-neuro endocrine relationships. Clin Immunol Immunopathol 18: 108–120.
Janković BD, Isaković K, Mićić M (1982) The thymus—hypophysis interaction in the developing chick embryo. Thymic epithelial cells in hypophysectomized embryos. In: Nienwenhuis vin den broek, Hana eds. In vivo Immunol. p. 343.
Juul A, Main K, Blum W, Lindholm J, Ranke M, Skakkebaek NE (1994) The ration between serum levels of insulin-like growth factor (IGF)-I and the IGF binding proteins (IGFBP-1, 2 and 3) decreases with age in healthy adults and is increased in agromegalic patients. Clin Endocrinol (Oxf) 41: 85–93.
Kalisnik M (1985) Temelji stereologije. Acta Stereol 4(Suppl. 1): Ljubljana.
Kaneshima H, Ito M, Asai J, Taguchi O (1987) Thymic epithelial reticular cell subpopulations in mice defined by monoclonal antibodies. Lab Invest 56: 372–380.
Karapetrovic B, Micic M, Leposavic G (1995) Stereological analysis of the sexually mature rat thymus after orchidectomy. Indian J Med Res 102: 42–48.
Knyszynski A, Adler-Kunin S, Globerson A (1992) Effects of growth hormone on thymocyte development from progenitor cells in the bone marrow. Brain Behav Immunol 6: 327–340.
Levite J, Cahalon L, Hershkoviz R, Steinman L, Lider O (1998) Neuropeptides, via specific receptors, regulate T-cell adhesion to fibronectin. J Immunol 160: 993–1000.
Li YM, Brunke DL, Dantzer R, Kelley KW (1992) Pituitary epithelial cell implants reverse the accumulation of CD4-CD8-lymphocytes in thymus glands of aged rats. Endocrinology 130: 2703–2709.
Lind EF, Prockop SE, Porritt HE, Petrie HT (2001) Mapping precursor movement through the postnatal thymus reveals specific microenvironments supporting defined stages of early lymphoid development. J Exp Med 194: 127–134.
Maggiano N, Plantelli M, Ricci R, Larocca LM, Capelli A, Ranelletti FO (1994) Detection of growth hormone-producing cells in human thymus by immunohistochemistry and non-radioactive in situ hybridization. J Histochem Cytochem 42: 1349–1354.
Martin A, Alonson L, Gomez Del Moral M, Zapata AG (1994) Morphometrical changes in the rat thymic lymphoid cells after treatment with two different doses of estradiol benzoate. Histol Histopath 9: 281–286.
Mascardo RN, Barton RW, Sherline P (1984) Somatostatin has an anti proliferative effect on concavalin-activated rat thymocytes. Clin Immunol Immunopathol 33: 131–138.
Moreno J, Varas A, Vicente A, Zapata AG (1998) Role of prolactin in the recovered T-cell development of early partially decapitated chicken embryo. Dev Immunol 5: 183–195.
Murphy WJ, Durum SK, Longo DL (1993) Differential effects of growth hormone and prolactin on murine T cell development and function. J Exp Med 178: 231–236.
Nakahama M, Mohri N, Mori S, Shindo G, Yokoi Y, Machinami R (1990) Immunohistochemical and histometrical studies on the human thymus with special emphasis on age-related changes in medullary epithelial and dendritic cells. Virchows Arch 58: 245–251.
Palmer DB, Hayday A, Owen MJ (1993) Is TCRβ expression an essential event in early thymocyte development? Immunol Today 14: 460–462.
Patel YC (1999) Somatostatin and its receptor family. Front Neuroendocrinol 20: 157–198.
Penit C, Lucas B, Vasseur F (1995) Cell expansion and growth arrest phases during the transition from precursor (CD4−8−) to immature (CD4+8+) thymocytes in normal and genetically modified mice. J Immunol 154: 5103–5113.
Pierpaoli W, Baroni C, Fabris N, Sorkin E (1969) Hormones and immunological capacity. II. Reconstitution of antibody production in hormonally deficient mice by somatotropic hormone, thyrotropic hormone, and thyroxin. Immunology 16: 217–230.
Reubi JC, Waser B, Horisberger U, Krenning E, Lamberts SWJ, Gebbers JO, Gersbach P, Laissue JA (1993) In vitro autoradiographic and in vivo scintigraphic localization of somatostatin receptors in human lymphatic tissue. Blood 82: 2143–2151.
Rouse RV, Bolin LM, Bender JR, Kyewski BA (1988) Monoclonal antibodies reactive with subsets of mouse and human thymic epithelial cells. J Histochem Cytochem 36: 1511–1517.
Sabharwal P, Varma S (1996) Growth hormone synthesized and secreted by human thymocytes acts via insulin-like growth factor 1 as an autocrine and paracrine growth factor. J Clin Endocrinol Metab 81: 2663–2669.
Savino W, Dardenne M (2000) Neuro-endocrine control of thymus physiology. Endocrine Rev 21: 12–443.
Serri O, Brazeau P, Kachra Z, Posner B (1992) Octreotide inhibits insulin-like growth factor-I hepatic gene expression in the hypophysectomized rat: Evidence for a direct and indirect mechanism of action. Endocrinology 130: 1816–1821.
Sharma K, Patel YC, Srikant CB (1996) Subtype selective induction of wild-type p53 and apoptosis, but not cell cycle arrest, by human somatostatin receptor 3. Mol Endocrinol 10: 1688–1696.
Smith P (1930) The effect of hypophysectomy upon the involution of the thymus in the rat. Anat Rec 47: 119–143.
Srikant CB (1995) Cell cycle dependent induction of apoptosis by somatostatin analog SMS 201-959 in AtT-20 mouse pituitary tumor cells. Biochem Biophys Res Commun 209: 400–407.
Starčević V, Morrow BA, Farner LA, Keil LC, Severs WB (1988) Longterm recording of cerebrospinal fluid pressure in freely behaving rats. Brain Res 462: 112–117.
Starr TK, Jameson SC, Hogquist KA (2003) Positive and negative selection of T cells. Annu Rev Immunol 21: 139–176.
Steinmann GG (1986) Changes in the human thymus during ageing. In: Muller-Hermelink HK, ed. Current Topics in Pathology. The Human Thymus. Berlin, Heidelberg: Springer-Verlag, pp. 43–88.
Timsit J, Savino W, Safieh B, Chanson P, Gagnerault MC, Bach JF, Dardenne M (1992) Growth hormone and insulin-like growth factor-1 stimulate hormonal function and proliferation of thymic epithelial cells. J Clin Endocrinol Metab 75: 183–188.
Van Vliet E, Melis M, van Ewijk W (1984) Monoclonal antibodies to stroma cell types in the mouse thymus. Eur J Immuol 14: 524–529.
Weibel ER (1979) Stereological Methods. Practical Methods for Biological Morphometry. London: Academic Press, pp. 1–415.
Wiedermann CJ, Renisch N, Braunsteiner H (1993) Stimulation of monocyte chemotaxis by human growth hormone and its deactivation by somatostatin. Blood 82: 954–960.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Petrovic-Djergovic, D.M., Rakin, A.K., Zivkovic, I.P. et al. Changes in the Thymus of Peripubertal Rats Induced by Centrally Applied Somatostatin-28. Histochem J 34, 573–582 (2003). https://doi.org/10.1023/A:1026047600493
Issue Date:
DOI: https://doi.org/10.1023/A:1026047600493