Abstract
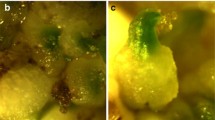
A rapid and efficient regeneration system has been developed for interspecific self-incompatible hybrids between Antirrhinum majus×A. hispanicum. Multiple shoots were induced from hypocotyls explants on Murashige and Skoog medium or Loblolly pine medium supplemented with a low concentration of 3-indoleacetic acid (0.05 mg l−1) and a high concentration of 6-benzyladenine (1 mg l−1). Rooting took place in 1/2 Murashige and Skoog medium without growth regulators. By using this procedure, it takes about 5–6 weeks from seed to plantlet. The hypocotyls also had a potential to expand and form callus, providing a source for a continuous supply of multiple shoots from the calli at periodic intervals.
Similar content being viewed by others
References
Atkinson NJ, Ford-Lloyd BV &; HJ Newbury (1989) Regeneration of plants from Antirrhinum majus L. callus. Plant Cell Tiss. Org. Cult. 17: 59–70
Bradley D, Carpenter R, Sommer H, Hartley N &; Coen E (1993) Complementary floral homeotic phenotypes result from opposite orientations of a transposon at the plena locus of Antirrhinum. Cell 72: 85–95
Coen ES (1992) Flower development. Curr. Opin. Cell Biol. 4: 929–933
Cui ML, Takayanagi K, Kamada H, Nishimura S &; Handa T (2001) Efficient shoot regeneration from hairy roots of Antirrhinum majus L. transformed by the rol type MAT vector system. Plant Cell Rep. 20: 55–59
González-Benito ME, Tapia J, Rodriguez N &; Iriondo JM (1996) Micropropagation of commercial and wild genotypes of snapdragon (Antirrhinum spp.). J. Hort. Sci. 71: 11–15
Heidmann I, Efremova N, Saedler H &; Schwarz-Sommer Z (1998) A protocol for transformation and regeneration of Antirrhinum majus. Plant J. 13: 723–728
Litany JD, Verma DC &; Johnson MA (1985) Influence of a loblolly pine (Pinus taeda L.). Culture medium and its components on growth and somatic embryogenesis of the wild carrot (Daucus carota L.). Plant Cell Rep. 4: 325–328
Luo D, Carpenter R, Copsey L, Vincent C, Clark J &; Coen E (1999) Control of organ asymmetry in flowers of Antirrhinum. Cell 99: 369–376
Murashige T &; Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 15: 473–497
Nath U, Crawford BC, Carpenter R &; Coen E (2003) Genetic control of surface curvature. Science 28: 1404–1407
Okubo H, Wada K &; Uemoto S (1991) In vitro morphogenetic response and distribution of endogenous plant hormones in hypocotyls segments of snapdragon (Antirrhinum majus L.). Plant Cell Rep.10: 501–504
Poirier-Hammon S, Rao PS &; Harada H (1974) Culture of mesophyll protoplasts and stem segments of Antirrhinum majus (snapdragon): growth and organization of embryoids. J. Exp. Bot. 87: 752–760
Pfister JM &; Widholm JM (1984) Plan regeneration from snapdragon tissue cultures. Hort Science 19: 852–854
Sangwan RS &; Harada H (1975) Chemical regulation of callus growth, organogenesis, plant regeneration, and somatic embryogenesis in Antirrhinum majus tissue and cell cultures. J. Exp. Bot. 26: 868–881
Xue Y, Carpenter R, Dickinson HG &; Coen ES (1996) Origin of allelic diversity in Antirrhinum S locus RNases. Plant Cell 8: 805–814
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, H., Ouyang, J. & Xue, Y. A facile procedure for efficient plantlet regeneration from self-incompatible hybrids in Antirrhinum . Plant Cell, Tissue and Organ Culture 76, 61–65 (2004). https://doi.org/10.1023/A:1025899532273
Issue Date:
DOI: https://doi.org/10.1023/A:1025899532273