Abstract
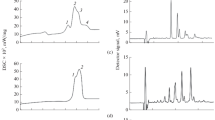
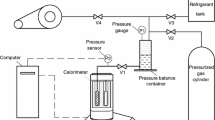
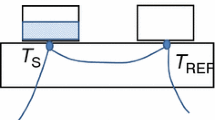
A simple method is proposed to estimate the vaporization enthalpy of the palmitic acid (hexadecanoic acid) at its normal boiling temperature. Differential scanning calorimetry (DSC) was the technique used to directly measure these thermodynamic properties. The advantages of this method are its speed and small amount of sample required. In order to avoid evaporation and to ensure equilibrium conditions, the experiments were carried out including a-alumina in contact with the fatty acid. The effect of the alumina concentration is discussed. The obtained experimental data (Tbp=625.4±0.5 K, Dvap H=237.6±5.9 J g-1) is compared with that obtained by using thermodynamic equations.
Similar content being viewed by others
References
R. Fuchs and W. K. Stephenson, Can. J. Chem., 63 (1985) 349; R. Fuchs, E. J. Chambers and W. K. Stephenson, Can. J. Chem., 65 (1987) 2624.
J. S. Chickos, S. Hosseini and D. G. Hesse, Thermochim. Acta, 249 (1995) 41.
M. A. V. Ribeiro da Silva, A. R. Matos and L. M. P. F. Amaral, J. Chem. Thermodyn., 27 (1995) 565.
F. A. Adedeji, D. L. S. Brown, J. A. Connor, M. Leung, M. I. Paz-Andrade and H. A. Skinner, J. Organomet. Chem., 97 (1975) 221.
M. A. Flamm-Ter Meer, H. D. Beckhaus and C. Rüchardt, Thermochim. Acta, 80 (1984) 81.
J. S. Chickos, D. G. Hesse, S. Hosseini, J. F. Liebman, G. D. Mendenhall, S. P. Verevkin, K. Rakus, H. D. Beckhaus and C. Rüchardt, J. Chem. Thermodyn., 27 (1995) 693.
J. S. Chickos and D. G. Hesse, J. Org. Chem., 54 (1989) 5250.
Z. Y. Liu, Chem. Eng. Comm., 184 (2001) 221.
P. J. Sánchez-Soto, J. M. Ginés, M. J. Arias, Cs. Novák and A. Ruiz-Conde, J. Therm. Anal. Cal., 67 (2002) 189.
A. Marini, V. Berbenni, G. Bruni, M. Villa and A. Orlandi, J. Therm. Anal. Cal., 68 (2002) 389.
V. T. Popa and E. Segal, J. Therm. Anal. Cal., 69 (2002) 149.
P. Juhász, J. Varga, K. Belina and H. Marand, J. Therm. Anal. Cal., 69 (2002) 561.
L. S. Semko, L. S. Dzyubenko, V. M. Ogenko and S. L. Revo, J. Therm. Anal. Cal., 70 (2002)
K. Mogi, H. Kubokawa and T. Hatakeyama, J. Therm. Anal. Cal., 70 (2002) 867.
M. Tsuchiya and T. Koyima, J. Therm. Anal. Cal., 72 (2003) 651.
S. Montserrat, F. Roman and P. Colomer, J. Therm. Anal. Cal., 72 (2003) 657
F. M. Etzler and J. J. Conners, Thermochim. Acta, 189 (1991) 185.
E. M. Barral, Thermochim. Acta, 5 (1973) 377.
X. Cia, D. C. Du and Y. M. Jin, J. Thermal Anal., 45 (1995) 193.
R. J. Seyler, Thermochim. Acta, 17 (1976) 129.
J. W. Goodrum and E. M. Siesel, J. Thermal Anal., 46 (1996) 1251.
J. W. Goodrum, D. P. Geller and S. A. Lee, Thermochim. Acta, 311 (1998) 71.
F. O. Cedeño, M. M. Prieto, A. Espina and J. R. García, Thermochim. Acta, 369 (2001) 39.
M. M. Prieto, J. C. Bada, M. I. Lombardía and E. Graciani, Grasas y Aceites, 49 (1998) 151.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cedeńo, F.O., Prieto, M.M., Espina, A. et al. Fast method for the experimental determination of vaporization enthalpy by differential scanning calorimetry. Journal of Thermal Analysis and Calorimetry 73, 775–781 (2003). https://doi.org/10.1023/A:1025878313429
Issue Date:
DOI: https://doi.org/10.1023/A:1025878313429