Abstract
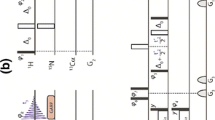
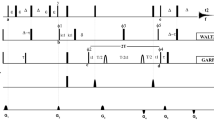
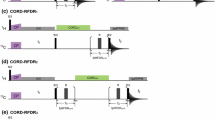
Novel NMR pulse schemes for simultaneous measurement of 1 D CαHαand 2 D NHαresidual dipolar couplings in proteins is presented. We show that 2 D NHαcoupling can be very useful for protein structure determination. The 2 D NHαcoupling can be measured from 15N dimension with good accuracy on a slowly relaxing TROSY resonance, utilizing HNCA-TROSY-based experiments, which concomitantly supply large 1 D CαHαcoupling. The dynamic range of 2 D NHαcoupling is comparable to 1 D NC′ coupling, but instead, it also serves non-redundant information on the course of protein backbone, thanks to rotational degree of freedom with respect to peptide bond. The HNCA-TROSY-based experiments are optimal for measuring residual dipolar couplings at high magnetic fields owing to absence of rapid transverse relaxation of carbonyl carbon. The reliability of the proposed approach was tested on 15N/13C human ubiquitin. A very good correlation with ubiquitin solution as well as crystal structure, for both 1 D CαHαand 2 D NHαcouplings, was obtained.
Similar content being viewed by others
References
Annila, A., Aitio, H., Thulin, E. and Drakenberg, T. (1999) J. Biomol. NMR, 14, 223-230.
Bax, A. and Tjandra, N. (1997) J. Biomol. NMR, 10, 289-292.
Bax, A., Kontaxis, G. and Tjandra, N. (2001) Meth. Enzymol., 339, 127-174.
Cai, M., Wang, H., Olejniczak, E.T., Meadows, R.P., Gunasekera, A.H., Xu, N. and Fesik, S. (1999) J. Magn. Reson., 139, 451-453.
Carlomagno, T., Peti, W. and Griesinger, C. (2000) J. Biomol. NMR, 17, 99-109.
Chou, J.J. and Bax, A. (2001) J. Am. Chem. Soc., 123, 3844-3845.
Clore, G.M., Gronenborn, A.M. and Tjandra, N. (1998a) J. Magn. Reson., 131, 159-162.
Clore, G.M., Gronenborn, A.M. and Bax, A. (1998b) J. Magn. Reson., 133, 216-221.
Cornilescu, G., Marquardt, J.L., Ottiger, M. and Bax, A. (1998) J. Am. Chem. Soc., 120, 6836-6837.
De Alba, E. and Tjandra, N. (2002) Prog. Nucl. Magn. Reson. Spectrosc., 40, 175-197.
Hansen, M.R., Mueller, L. and Pardi, A. (1998) Nat. Struct. Biol., 5, 1065-1074.
Kaikkonen, A. and Otting, G. (2001) J. Am. Chem. Soc., 123, 1770-1771.
Kay, L.E., Keifer, P. and Saarinen, T. (1992) J. Am. Chem. Soc., 114, 10663-10665.
Kontaxis, G. and Bax, A. (2001) J. Biomol. NMR, 20, 77-82.
Logan, T.M., Olejniczak, E.T., Xu, X.R. and Fesik, S.W. (1993) J. Biomol. NMR, 3, 225-231.
Loria, J.P., Rance, M. and Palmer III, A.G. (1999) J. Magn. Reson., 141, 180-184.
Losonczi, J.A., Andrec, M., Fischer, M.W.F. and Prestegard, J.H. (1999) J. Magn. Reson., 138, 334-342.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989) J. Magn. Reson., 85, 393-399.
McCoy, M. and Mueller, L. (1992) J. Am. Chem. Soc., 114, 2108-2112.
Meier, S., Hässinger, D., Jensen, P., Rogowski, M. and Grzesiek, S. (2003) J. Am. Chem. Soc., 125, 44-45.
Meissner, A., Duus, J.Ø. and Sørensen, O.W. (1997) J. Biomol. NMR, 10, 89-94.
Mittermaier, A. and Kay, L.E., (2001) J. Am. Chem. Soc., 123, 6892-6903.
Ottiger, M., Delaglio, F. and Bax, A. (1998a) J. Magn. Reson., 131, 373-378.
Ottiger, M., Delaglio, F., Marquardt, J.L., Tjandra, N. and Bax, A. (1998b) J. Magn. Reson., 134, 365-369.
Otting, G., Rückert, M., Levitt, M.H. and Moshref, A. (2000) J. Biomol. NMR, 16, 343-346.
Permi, P. (2001) J. Magn. Reson., 153, 267-272.
Permi, P. (2002a) J. Biomol. NMR, 22, 27-35.
Permi, P. (2002b) J. Biomol. NMR, 23, 201-209.
Permi, P. and Annila, A. (2000) J. Biomol. NMR, 16, 221-227.
Permi, P. and Annila, A. (2001b) J. Biomol. NMR, 20, 127-133.
Permi, P. and Annila, A. (2002) J. Magn. Reson., 155, 123-130.
Permi, P., Heikkinen, S, Kilpeläinen, I. and Annila, A. (1999) J. Magn. Reson., 139, 273-280.
Permi, P., Rosevear, P.R. and Annila, A. (2000a) J. Biomol. NMR, 17, 43-54.
Permi, P., Kilpeläinen, I. and Annila, A. (2000b) J. Magn. Reson., 146, 255-259.
Pervushin, K., Riek, R., Wider, G. and Wüthrich, K. (1997) Proc. Natl. Acad. Sci. USA, 94, 12366-12371.
Prestegard, J.H., Al-Hashimi, H.M. and Tolman, J.R. (2000) Quart. Rev. Biophys., 33, 371-424.
Salzmann, M., Pervushin, K., Wider, G., Senn, H. and Wüthrich, K. (1998) Proc. Natl. Acad. Sci. USA, 15, 181-185.
Seip, S., Balbach, J. and Kessler, H. (1992) Angew. Chem., 104, 1656-1658.
Tian, F., Valafar, H. and Prestegard, J.H. (2001) J. Am. Chem. Soc., 123, 11791-11796.
Tjandra, N. and Bax, A. (1997) Science, 278, 1111-1114.
Tolman, J.R., Flanagan, J.M., Kennedy, M.A. and Prestegard, J.H. (1995) Proc. Natl. Acad. Sci. USA, 92, 9279-9283.
Vijay-Kumar, S., Bugg, C.E. and Cook, W.J. (1987) J. Mol. Biol., 194, 531-544.
Vögeli, B. and Pervushin, K. (2002) J. Biomol. NMR, 24, 291-300.
Wand, A.C. and Bax, A. (1996) J. Am. Chem. Soc., 118, 2483-2494.
Weigelt, J. (1998) J. Am. Chem. Soc., 120, 10778-10779.
Wu, Z. and Bax, A. (2002) J. Am. Chem. Soc., 124, 9672-9673.
Yang, D. and Nagayma, K. (1996) J. Magn. Reson., A118, 117-121.
Yang, D. and Kay, L.E. (1999) J. Biomol. NMR, 14, 273-276.
Yang, D., Tolman, J.R., Goto, N.K. and Kay, L.E. (1998) J. Biomol. NMR, 12, 325-332.
Yang, D., Venters, R.A., Mueller, G.A., Choy, W.Y. and Kay, L.E. (1999) J. Biomol. NMR, 14, 333-343.
Zweckstetter, M. and Bax, A. (2000) J. Am. Chem. Soc., 122, 3791-3792.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Permi, P. Measurement of residual dipolar couplings from 1Hα to 13Cα and 15N using a simple HNCA-based experiment. J Biomol NMR 27, 341–349 (2003). https://doi.org/10.1023/A:1025866606252
Issue Date:
DOI: https://doi.org/10.1023/A:1025866606252