Abstract
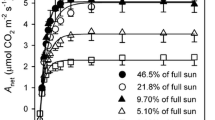
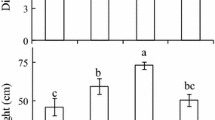
Twelve-year-old Norway spruce (Picea abies [L.] Karst.) were exposed to ambient (AC) or elevated (EC) [ambient + 350 μmol(CO2) mol−1] CO2 concentration [CO2] using the facilities of open-top-chambers (OTCs) and glass domes (GDs). A combination of gas exchange measurements and application of a biochemical model of photosynthesis were used for the evaluation of CO2 assimilation characteristics. Morphological change was assessed on the base of specific leaf area (SLA). Nitrogen (N) content in the assimilation apparatus was considered a main factor influencing the biochemical capacity. Three experiments confirm the hypothesis that an adjustment of photosynthetic capacity under EC is controlled by the combination of biochemical, morphological, and physiological feedback mechanisms. We observed periodicity of down-regulation of photosynthetic capacity (Experiment No. 1) during the vegetation seasons. In the spring months (May–June), i.e. during the occurrence of active carbon sink associated with the formation of new foliage, up-regulation (10–35 %) of photosynthetic capacity (P Nsat) was observed. On the contrary, in the autumn months (September–October) down-regulation (25–35 %) of P Nsat was recorded that was mainly associated with reduced carbon sink strength and biochemical change, i.e. decrease of N status (up to 32 %) and accumulation of saccharides (up to 72 %) in leaves. Different adjustments of photosynthetic activities were observed in current (C) and one-year-old (C-1) needles exposed to EC (Experiment No. 2). Strong down-regulation of P Nsat and the diminution of the initial stimulation of photosynthetic rate (P Nmax) was associated with decreases of both ribulose-1,5-bisphosphate carboxylase/oxygenase carboxylation activity (by 32 %) and RuBP regeneration (by 40 %). This performance was tightly correlated with the absence of active carbon sinks, decrease of N content, and starch accumulation in C-1 needles. Finally, different responses of sun- and shade-adapted needles to EC (Experiment No. 3) were associated with the balance between morphological and biochemical changes. Observed P Nsat down-regulation (by 22 %) of exposed needles in EC was predominantly caused by effects of both higher assimilate accumulation and stronger N dilution, resulting from higher absolute photosynthetic rates and incident irradiances in the upper canopy.
Similar content being viewed by others
References
Ceulemans, R.: Direct impacts of CO2 and temperature on physiological processes in trees.-In: Mohren, G.M.J., Kramer, K., Sabaté, S. (ed.): Impacts of Global Change on Tree Physiology and Forest Ecosystems. Pp. 3-14. Kluwer Academic Publishers, Dordrecht-Boston-London 1997.
Ceulemans, R., Mousseau, M.: Effects of elevated atmospheric CO2 on woody plants.-New Phytol. 127: 425-446, 1994.
DeLucia, E.H., Sasek, T.W., Strain, B.R.: Photosynthetic inhibition after long-term exposure to elevated levels of atmospheric carbon dioxide.-Photosynth. Res. 7: 175-184, 1985.
Eamus, D., Jarvis, P.G.: The direct effects of increase in the global atmospheric CO2 concentration on natural and commercial temperate trees and forests.-In: Begon, M., Fitter, A.H., Ford, E.D., MacFadyen, A. (ed.): Advances in Ecological Research. Pp. 1-55. Academic Press, London-Tokyo-Toronto 1989.
Farquhar, G.D., Caemmerer, S. von, Berry, J.A.: A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species.-Planta 149: 78-90, 1980.
Gilmore, D.W., Seymour, R.S., Halteman, W.A., Greenwood, M.S.: Canopy dynamics and the morphological development of Abies balsamea: effects of foliage age on specific leaf area and secondary vascular development.-Tree Physiol. 15: 47-55, 1995.
Janouš, D., Dvořák, V., Opluštilová, M., Kalina, J.: Chamber effects and responses of trees in the experiment using open top chambers.-J. Plant Physiol. 148: 332-338, 1996.
Kalina, J., Urban, O., Čajánek, M., Kurasová, I., Špunda, V., Marek, M.V.: Different responses of Norway spruce needles from shaded and exposed crown layers to the prolonged exposure to elevated CO2 studied by various chlorophyll a fluorescence techniques.-Photosynthetica 39: 369-376, 2001.
Kerstiens, G.: Shade-tolerance as a predictor of responses to elevated CO2 in trees.-Physiol. Plant. 102: 472-480, 1998.
Kubiske, M.E, Pregitzer, K.S.: Ecophysiological responses to simulated canopy gaps of two tree species of contrasting shade tolerance in elevated CO2.-Funct. Ecol. 11: 24-32, 1997.
Loach, K., Little, C.H.A.: Production, storage, and use of photosynthate during shoot elongation in basal fir (Abies balsamea).-Can. J. Bot. 51: 1161-1168, 1973.
Luo, Y., Field, C.B., Mooney, H.A.: Predicting responses of photosynthesis and root fraction to elevated [CO2]a: Interactions among carbon, nitrogen, and growth: theoretical paper.-Plant Cell Environ. 17: 1195-1204, 1994.
Luo, Y., Reynolds, J., Wang, Y., Wolfe, D.: A search for predictive understanding of plant responses to elevated [CO2].-Global Change Biol. 5: 143-156, 1999.
Marek, M.V., Kalina, J., Matoušková, M.: Response of photosynthetic carbon assimilation of Norway spruce exposed to long-term elevation of CO2 concentration.-Photosynthetica 31: 209-220, 1995.
Marek, M.V., Šprtová, M., Kalina, J.: The photosynthetic irradiance-response of Norway spruce exposed to a long-term elevation of CO2 concentration.-Photosynthetica 33: 259-268, 1997.
Marek, M.V., Urban, O., Šprtová, M., Pokorný, R., Rosová, Z., Kulhavý, J.: Photosynthetic assimilation of sun versus shade Norway spruce [Picea abies (L.) Karst] needles under long-term impact of elevated CO2 concentration.-Photosynthetica 40: 259-267, 2002.
Martin, T. Oswald, O., Graham, I.A.: Arabidopsis seedling growth, storage lipid mobilization, and photosynthetic gene expression are regulated by carbon: nitrogen availability.-Plant Physiol. 128: 472-481, 2002.
Opluštilová, M., Dvořák, V.: Growth processes of Norway spruce in elevated CO2 concentration.-In: Mohren, G.M.J., Kramer, K., Sabaté, S. (ed.): Impacts of Global Change on Tree Physiology and Forest Ecosystems. Pp. 53-58. Kluwer Academic Publishers, Dordrecht 1997.
Osborne, C.P., LaRoche, J., Garcia, R.L., Kimball, B.A., Wall, G.W., Pinter, P.J., Jr., LaMorte, R.L., Hendrey, G.R., Long, S.P.: Does leaf position within a canopy affect acclimation of photosynthesis to elevated CO2? Analysis of a wheat crop under free-air CO2 enrichment.-Plant Physiol. 117: 1037-1045, 1998.
Overdieck, D., Kellomäki, S., Wang, K.Y.: Do the effects of temperature and CO2 interact?-In: Jarvis, P.G. (ed.): European Forests and Global Change. The Likely Impacts of Rising CO2 and Temperature. Pp. 236-273. Cambridge University Press, Cambridge 1998.
Pons, T.L., van Rijnberk, H., Scheurwater, I., van der Werf, A.: Importance of the gradient in photosynthetically active radiation in a vegetation stand for leaf nitrogen allocation in two monocotyledons.-Oecologia 95: 416-424, 1993.
Rey, A., Jarvis, P.G.: Long-term photosynthetic acclimation to increased atmospheric CO2 concentration in young birch (Betula pendula) trees.-Tree Physiol. 18: 441-450. 1998.
Roberntz, P., Stockfors, J.: Effects of elevated CO2 concentration and nutrition on net photosynthesis, stomatal conductance and needle respiration of field-grown Norway spruce trees.-Tree Physiol. 18: 233-241, 1998.
Sage, R.F.: A model describing the regulation of ribulose-1.5-bisphosphate carboxylase, electron transport, and triose phosphate use in response to light intensity and CO2 in C3 plants.-Plant Physiol. 94: 1728-1734, 1990.
Sage, R.F., Sharkey, T.D., Seemann, J.R.: Acclimation of photosynthesis to elevated CO2 in five C3 species.-Plant Physiol. 89: 590-596, 1989.
Saralabai, V.C., Vivekandan, M., Babu, R.S.: Plant responses to high CO2 concentration in the atmosphere.-Photosynthetica 33: 7-37, 1997.
Šesták, Z.: Leaf ontogeny and photosynthesis.-In: Johnson, C.B. (ed.): Physiological Processes Limiting Plant Productivity. Pp. 147-158. Butterworths, London-Boston-Sydney-Wellington-Durban-Toronto 1981.
Špunda, V., Kalina, J., Čajánek, M., Pavlíčková, H., Marek, M.V.: Long-term exposure of Norway spruce to elevated CO2 concentration induces changes in photosystem II mimicking an adaptation to increased irradiance.-J. Plant Physiol. 152: 413-419, 1998.
Stitt, M., Krapp, A.: The interaction between elevated carbon dioxide and nitrogen nutrition: the physiological and molecular background.-Plant Cell Environ. 22: 583-621, 1999.
Tissue, D.T., Griffin, K.L., Ball, J.T.: Photosynthetic adjustment in field-grown ponderosa pine trees after six years of exposure to elevated CO2.-Tree Physiol. 19: 221-228, 1999.
Tissue, D.T., Griffin, K.L., Turnbull, M.H., Whitehead, D.: Canopy position and needle age affect photosynthetic response in field-grown Pinus radiata after five years of exposure to elevated carbon dioxide partial pressure.-Tree Physiol. 21: 915-923, 2001.
Urban, O.: Physiological impacts of elevated CO2 concentration ranging from molecular to whole plant responses.-Photosynthetica 41: 9-20, 2003.
Urban, O., Janouš, D., Pokorný, R., Marková, I., Pavelka, M., Fojtík, Z., Šprtová, M., Kalina, J., Marek, M.V.: Glass domes with adjustable windows: A novel technique for exposing juvenile forest stands to elevated CO2 concentration.-Photosynthetica 39: 395-401, 2001.
Urban, O., Marek, M.V.: Seasonal changes of selected parameters of CO2 fixation biochemistry of Norway spruce under the long-term impact of elevated CO2.-Photosynthetica 36: 533-545, 1999.
Urban, O., Pokorný, R., Šalanská, P.: Seasonal dynamics of specific leaf area and specific branch area in Picea abies trees.-Zpravodaj Beskydy 13: 157-160, 2000.
Vu, J.C.V., Allen, L.H., Jr., Bowes, G.: Leaf ultrastructure, carbohydrates and protein of soybeans grown under CO2 enrichment.-Environ. exp. Bot. 29: 141-147, 1989.
Watson, R.T., Noble, I.R., Bolin, B., Ravindranath, N.H., Verardo, D.J., Dokken, D.J.: Land Use, Land-Use Change, and Forestry. A Special Report of the IPCC.-Pp. 3-5. Cambridge University Press, Cambridge 2000.
Webber, A.N., Nie, G.-Y., Long, S.P.: Acclimation of photosynthetic proteins to rising atmospheric CO2.-Photosynth. Res. 39: 413-425, 1994.
Wolfe, D.W., Gifford, R.M., Hilbert, D., Luo, Y.: Integration of photosynthetic acclimation to CO2 at the whole-plant level.-Global Change Biol. 4: 879-893, 1998.
Wullschleger, S.D., Norby, R.J., Hendrix, D.L.: Carbon exchange rates, chlorophyll content, and carbohydrate status of two forest tree species exposed to carbon dioxide enrichment.-Tree Physiol. 10: 21-31, 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Urban, O., Pokorný, R., Kalina, J. et al. Control Mechanisms of Photosynthetic Capacity Under Elevated CO2 Concentration: Evidence from Three Experiments with Norway Spruce Trees. Photosynthetica 41, 69–75 (2003). https://doi.org/10.1023/A:1025808428684
Issue Date:
DOI: https://doi.org/10.1023/A:1025808428684