Abstract
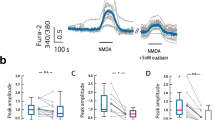
NMDA-induced modification of postsynaptic densities (PSDs) was studied by immunoelectron microscopy. Treatment of cultured hippocampal neurons with NMDA for 2 min promotes a 2.3 fold thickening of the PSD and a 4 fold increase in PSD-associated CaMKII immunolabel. These changes are reversed 5 min after the removal of NMDA and Ca2+ from the medium. In addition, following NMDA treatment, PSDs exhibit a 7.5 fold increase in labeling with an antibody specific to the (Thr286) phospho-form of CaMKII, indicating that CaMKII translocated to the PSD is phosphorylated. When the phosphatase inhibitors, calyculin A or okadaic acid, are included in the medium, the NMDA-induced thickening of the PSD as well as the increase in PSD-associated CaMKII immunolabeling are largely maintained (75% and 88% of the peak values respectively) at 5 min after removal of NMDA and Ca2+ from the medium. These results imply that NMDA receptors can mediate activity-induced changes in the PSD and that phosphatases of type 1 and/or 2A are involved in the reversal of these changes.
Similar content being viewed by others
References
Allen, P. B., Ouimet, C. C. & Greengard, P. (1997) Spinophilin, a novel protein phosphatase 1 binding protein localized to dendritic spines. Proceedings of the National Academy of Sciences of the USA 94, 9956–9961.
Bayer, K. U., de Koninck, P., Leonard, A. S., Hell, J. W. & Schulman, H. (2001) Interaction with theNMDAreceptor locks CaMKII in an active conformation. Nature 411, 801–805.
Bear, M. F. & Malenka, R. C. (1994) Synaptic plasticity: LTP and LTD. Current Opinion in Neurobiology 4, 389–399.
Cohen, P. (1989) The structure and regulation of protein phosphatases. Annual Review of Biochemistry 58, 453–508.
Dosemeci, A. & Choi, C. (1997) Ca(2+)-independent autophosphorylation of postsynaptic density-associated Ca2+/calmodulin-dependent protein kinase. Neurochemical Research 22, 1151–1157.
Dosemeci, A. & Reese, T. S. (1993) Inhibition of endogenous phosphatase in a postsynaptic density fraction allows extensive phosphorylation of the major postsynaptic density protein. Journal of Neurochemistry 61, 550–555.
Dosemeci, A., Reese, T. S., Petersen, J. & Tao-Cheng, J. H. (2000) A novel particulate form of Ca(2+)/calmodulin-dependent [correction of Ca(2+)/CaMKII-dependent] protein kinase II in neurons. Journal of Neuroscience 20, 3076–3084.
Dosemeci, A., Tao-cheng, J. H., Vinade, L., Winters, C. A., Pozzo-Miller, L. & Reese, T. S. (2001) Glutamate-induced transient modification of the postsynaptic density. Proceedings of the National Academy of Sciences of the USA 98, 10428–10432.
Hu, B. R., Park, M., Martone, M. E., Fischer, W. H., Ellisman, M. H. & Zivin, J. A. (1998) Assembly of proteins to postsynaptic densities after transient cerebral ischemia. Journal of Neuroscience 18, 625–633.
Ishihara, H., Martin, B. L., Brautigan, D. L., Karaki, H., Ozaki, H., Kato, Y., Fusetani, N., Watabe, S., Hashimoto, K. & Uemura, D. (1989) CalyculinAand okadaic acid: Inhibitors of protein phosphatase activity. Biochemical & Biophysical Research Communications 159, 871–877.
Lisman, J. (1989) A mechanism for the Hebb and the anti-Hebb processes underlying learning and memory. Proceedings of the National Academy of Sciences of the USA 86, 9574–9578.
Marrs, G. S., Green, S. H. & Dailey, M. E. (2001) Rapid formation and remodeling of postsynaptic densities in developing dendrites. Nature Neuroscience 4, 1006–1013.
Martone, M. E., Jones, Y. Z., Young, S. J., Ellisman, M. H., Zivin, J. A. & Hu, B. R. (1999) Modification of postsynaptic densities after transient cerebral ischemia: A quantitative and threedimensional ultrastructural study. Journal of Neuroscience 19, 1988–1997.
Ouimet, C. C., da Cruz e Silva, E. F. & Greengard, P. (1995) The alpha and gamma 1 isoforms of protein phosphatase 1 are highly and specifically concentrated in dendritic spines. Proceedings of the National Academy of Sciences of the USA 92, 3396–3400.
Shen, K. & Meyer, T. (1999) Dynamic control of CaMKII translocation and localization in hippocampal neurons by NMDA receptor stimulation. Science 284, 162–166.
Shen, K., Teruel, M. N., Connor, J. H., Shenolikar, S. & Meyer, T. (2000) Molecular memory by reversible translocation of calcium/calmodulin-dependent protein kinase II. Nature Neuroscience 3, 881–886.
Sheng, M. & Kim, M. J. (2002) Postsynaptic signaling and plasticity mechanisms. Science 298, 776–780.
Shenolikar, S. & Nairn, A. C. (1991) Protein phosphatases: Recent progress. Advances in Second Messenger and Phosphoprotein Research 23, 1–121.
Strack, S., Barban, M. A., Wadzinski, B. E. & Colbran, R. J. (1997) Differential inactivation of postsynaptic density-associated and soluble Ca2+/calmodulin-dependent protein kinase II by protein phosphatases 1 and 2A. Journal of Neurochemistry 68, 2119–2128.
Strack, S. & Colbran, R. J. (1998) Autophosphorylation-dependent targeting of calcium/calmodulindependent protein kinase II by the NR2B subunit of the N-methyl-D-aspartate receptor. Journal of Biological Chemistry 273, 20689–20692.
Strack, S., Kini, S., Ebner, F. F., Wadzinski, B. E. & Colbran, R. J. (1999) Differential cellular and subcellular localization of protein phosphatase 1 isoforms in brain. Journal of Comparative Neurology 413, 373–384.
Suzuki, T., Okumura-Noji, K., Tanaka, R. & Tada, T. (1994) Rapid translocation of cytosolic Ca2+/calmodulin-dependent protein kinase II into postsynaptic density after decapitation. Journal of Neurochemistry 63, 1529–1537.
Tomita, S., Nicoll, R. A. & Bredt, D. S. (2001) PDZ protein interactions regulating glutamate receptor function and plasticity. Journal of Cell Biology 153, F19–24.
Yen, L. H., Sibley, J. T. & Constantine-Paton, M. (1993) Fine-structural alterations and clustering of developing synapses after chronic treatments with low levels of NMDA. Journal of Neuroscience 13, 4949–4960.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dosemeci, A., Vinade, L., Winters, C.A. et al. Inhibition of phosphatase activity prolongs NMDA-induced modification of the postsynaptic density. J Neurocytol 31, 605–612 (2002). https://doi.org/10.1023/A:1025735410738
Issue Date:
DOI: https://doi.org/10.1023/A:1025735410738