Abstract
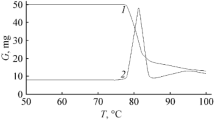
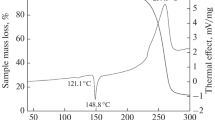
The thermal decomposition of 3-nitro-1-nitromethyl-1,2,4-1H-triazole in 1% solution in phenyl benzoate proceeds homolytically with initial rupture of the CH2-NO2 bond. Activation parameters of the process were E a = 172.6 kJ/mol, log A = 14.25. The initial basic pathway of fragmentation of the molecule under electron impact coincides with the first step of thermal decomposition, which is in agreement with X-ray structural and calculated quantum chemical data on bond stability in the molecule.
Similar content being viewed by others
REFERENCES
G. B. Manelis,G. M. Nazin,Yu. I. Rubtsov, and V. A. Strunin, Thermal Decomposition and Combustion of Explosive Substances and Powders [in Russian], Nauka, Moscow (1996).
A. D. Vasiliev,A. M. Astachov,O. A. Golubtsova,L. A. Kruglyakova, and R. S. Stepanov, Acta Crystallogr. C, 56, 999 (2000).
Yu. V. Shurukhin,N. A. Klyuev, and I. I. Grand berg, Khim. Geterotsikl. Soedin., 723 (1985).
R. S. Stepanov and L. A. Kruglyakova, Kinet. Katal., 37, 339 (1996).
R. S. Stepanov, Physicochemical Testing of Explosive Substances, Vol. 1 [in Russian], Izd. Krasnoyarsk. Politekh. Inst., Krasnoyarsk (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stepanov, R.S., Kruglyakova, L.A., Golubtsova, O.A. et al. Thermal Decomposition of 3-Nitro-1-nitromethyl-1,2,4-1H-triazole in Solution. Chemistry of Heterocyclic Compounds 39, 604–607 (2003). https://doi.org/10.1023/A:1025142015046
Issue Date:
DOI: https://doi.org/10.1023/A:1025142015046