Abstract
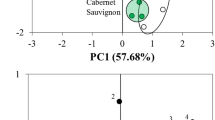
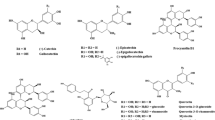
Flavonoids are a group of naturally occurring antioxidant compounds found in wine that are thought to have therapeutic importance in cardiovascular disease [1, 2]. The flavonoid content of red wines can differ as a function of the variety of wine examined [3–7]. Since there is a paucity of data on the content of these antioxidants in Merlot wine, we used high performance liquid chromatography to identify and compare catechin, epicatechin, rutin, transresveratrol and quercetin levels in selected Merlot wines from Canada, Chile and the United States. Additionally, antioxidant content was correlated with the price of the wine. Catechin content was the most abundant when compared to the other four phenolic compounds. The concentrations of each compound in the Merlot wines also varied as a function of the country of origin. Catechin and transresveratrol occurred in significantly lower concentrations in Merlots from the United States. The lowest levels of rutin were observed in Canadian Merlots. Quercetin occurred at significantly higher levels in Chilean Merlots. Wine prices were inversely correlated with catechin concentration. Merlot wine represents a source of antioxidants that may have an impact on cardiovascular disease.
Similar content being viewed by others
References
Riemersma RA, Rice-Evans CA, Tyrell RM, Clifford MN, Lean MEJ: Tea flavonoids and cardiovascular health. Q J Med 94: 277-282, 2001
Rimm EB, Katan MB, Ascherio A, Stampfer MJ, Willett WC: Relation between intake of flavonoids and risk for coronary heart disease in male health Professionals. Ann Intern Med 125: 384-389, 1996
Kopp P: Resveratrol, a phytoestrogen found in red wine. A possible explanation for the conundrum of the 'French paradox'? Eur J Endocrinol 138: 619-620, 1998
Nigdikar SV, Williams NR, Griffin BA, Howard AN: Consumption of red wine polyphenols reduces the susceptibility of low-density lipoproteins to oxidation in vivo. Am J Clin Nutr 68: 258-265, 1998
Iijima K, Yoshizumi M, Hashimoto M, Kim S, Eto M, Ako J, Liang Y-Q, Sudoh N, Hosoda K, Nakahara K, Toba K, Ouchi Y: Red wine polyphenols inhibit proliferation of vascular smooth muscle cells and downregulate expression of cyclin A gene. Circulation 101: 805-811, 2000
Abu-Amsha R, Croft KD, Puddey IB, Proudfoot JM, Beilin LJ: Phenolic content of various beverages determines the extent of inhibition of human serum and low-density lipoprotein oxidation in vitro: Identification and mechanism of action of some cinnamic acid derivatives from red wine. Clin Sci 91: 449-458, 1996
Kerry NL, Abbey M: Red wine and fractionated phenolic compounds prepared from red wine inhibit low density lipoprotein oxidation in vitro. Atherosclerosis 135: 93-102, 1997
Steinberg D: A critical look at the evidence for the oxidation of LDL in atherogenesis. Atherosclerosis 131(suppl): S5-S7, 1997
Berliner JA, Navab M, Fogelman AM, Frank JS, Demer LL, Edwards PA, Watson AD, Lusis AJ: Atherosclerosis: Basic mechanisms. Oxidation, inflammation and genetics. Circulation 91: 2488-2496, 1995
Giugliano D: Dietary antioxidants for cardiovascular prevention. Nutr Metab Cardiovasc Dis 10: 38-44, 2000
Frei B: Molecular and biological mechanisms of antioxidant action. FASEB J 13: 963-964, 1999
Chopra M, Thurnham DI: Antioxidants and lipoprotein metabolism. Proc Nutr Soc 58: 663-671, 1999
Renaud S, Lorgeril MD: Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet 339: 1523-1526, 1992
Teissedre PL, Frankel EN, Waterhouse AL, Peleg H, German JB: Inhibition of in vitro human LDL oxidation by phenolic antioxidants from grapes and wines. J Sci Food Agric 70: 55-61, 1996
Miyagi Y, Miwa K, Inoue H: Inhibition of human low-density lipoprotein oxidation by flavonoids in red wine and grape juice. Am J Cardiol 80: 1627-1631, 1997
deWhalley CV, Rankin SM, Hoult JRS, Jessup W, Leake DS: Flavonoids inhibit the oxidative modification of low density lipoproteins by macrophages. Biochem Pharmacol 39: 1743-1750, 1990
Gilly R, Mara D, Oded S, Zohar K: Resveratrol and a novel tyrosinase in carignan grape juice. J Agric Food Chem 49: 1479-1485, 2001
Pace-Asciak CR, Hahn S, Diamandis EP, Soleas G, Goldberg DM: The red wine phenolics trans-resveratrol and quercetin block human platelet aggregation and eicosanoid synthesis: Implications for protection against coronary heart disease. Clin Chim Acta 207-219, 1995
Pendurthi UR, Williams JT, Rao LVM: Resveratrol, a polyphenolic compound found in wine, inhibits tissue factor expression in vascular cells. Vasc Biol 19: 419-426, 1999
Rotondo S, Rajtar G, Manarini S, Celardo A, Rotilio D, Gaetano GD, Evangelista V, Cerletti C: Effect of trans-resveratrol, a natural polyphenolic compound, on human polymorphonuclear leukocyte function. Br J Pharmacol 123: 1691-1699, 1998
El-Mowafy AM, White RE: Resveratrol inhibits MAPK activity and nuclear translocation in coronary artery smooth muscle: Reversal of endothelin-1 stimulatory effects. FEBS Lett 451: 63-67, 1999
Stojanovic S, Sprinz H, Brede O: Efficiency and mechanism of the antioxidant action of trans-resveratrol and its analogues in the radical liposome oxidation. Arch Biochem Biophys 391: 79-89, 2001
Wu J, Wang Z, Hsieh T, Bruder J, Zou J, Huang Y: Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine. Int J Mol Med 8: 3-17, 2001
Frémont L, Belguendouz L, Delpal S: Antioxidant activity of resveratrol and alcohol-free wine polyphenols related to LDL oxidation and polyunsaturated fatty acids. Life Sci 64: 2511-2521, 1999
Landrault N, Poucheret P, Ravel P, Gasc F, Cros G, Teissedre P-L: Antioxidant capacities and phenolics levels of French wines from different varieties and vintages. J Agric Food Chem 49: 3341-3348, 2001
Goldberg DM, Tsang E, Karumanchiri A, Diamandis EP, Soleas G, Ng E: Method to assay the concentrations of phenolic constituents of biological interest in wines. Anal Chem 68: 1688-1694, 1996
Ghiselli A, Nardini M, Baldi A, Scaccini C: Antioxidant activity of different phenolic fractions separated from an Italian red wine. J Agric Food Chem 46: 361-367, 1998
Lamuela-Raventós RM, Waterhouse AL: A direct HPLC separation of wine phenolics. Am J Enol Vitic 45: 1-5, 1994
Roggero J-P, Archier P, Coen S: Wine phenolics analysis via direct injection: Enhancement of the method. J Liq Chromatogr 14: 533-538, 1991
Roggero J-P, Coen S, Archier P: Wine phenolics: Optimization of HPLC analysis. J Liq Chromatogr 13: 2593-2603, 1990
Salagoïty-Auguste M-H, Bertrand A: Wine phenolics-analysis of low molecular weight components by high performance liquid chromatography. J Sci Food Agric 35: 1241-1247, 1984
Arts ICW, Putte Bvd, Hollman PCH: Catechin contents of foods commonly consumed in the Netherlands. 2. Tea, wine, fruit juices, and chocolate milk. J Agric Food Chem 48: 1752-1757, 2000
Jeandet P, Bessis R, Sbaghi M, Meunier P, Trollat P: Resveratrol content of wines of different ages: Relationship with fungal disease pressure in the vineyard. Am J Enol Vitic 46: 1-4, 1995
Siemann EH, Creasy LL: Concentration of the phytoalexin resveratrol in wine. Am J Enol Vitic 43: 49-52, 1992
Makris DP, Rossiter JT: Comparison of quercetin and a non-orthohydroxy flavonol as antioxidants by competing in vitro oxidation reactions. J Agric Food Chem 49: 2001
Auw JM, Blanco V, O'Keefe SF, Sims CA: Effect of processing on the phenolics and color of Cabernet Sauvignon, Chambourcin, and Noble wines and juices. Am J Enol Vitic 47: 279-286, 1996
Martinez-Ortega MV, Carcia-Parrilla MC, Troncoso AM: Resveratrol content in wines and musts from the south of Spain. Nahrung 44: 253-256, 2000
Del Alamo M, Bernal JL, Gómez-Cordovés C: Behaviour of monosaccharides, phenolic compounds, and color of red wines aged in used oak barrels and in the bottle. J Agric Food Chem 48: 4613-4618, 2000
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Faustino, R., Sobrattee, S., Edel, A. et al. Comparative analysis of the phenolic content of selected Chilean, Canadian and American Merlot red wines. Mol Cell Biochem 249, 11–19 (2003). https://doi.org/10.1023/A:1024745513314
Issue Date:
DOI: https://doi.org/10.1023/A:1024745513314