Abstract
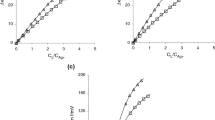
It is shown, using the formation of silver(I) pyridine (Py) and piperidine (Ppd) complexes in a di-methyl sulfoxide–acetonitrile (DMSO–AN) mixture with different compositions, that an increase in the stability of the complexes with an increase in the acetonitrile content is apparent and caused by the dilution factor. This follows from the more detailed specification of the chemical forms in a mixture of solvents, according to which the complexation is considered as the replacement of the DMSO molecules in the Ag(DMSO)+ 2 solvate ion by the ligands (Py or Ppd molecules). The Ag(DMSO)+ 2 form is more preferential than Ag(AN)+ 2 due to the stronger donor properties of DMSO.
Similar content being viewed by others
References
Fedorov, V.A. and Belevantsev, V.I., Abstracts of Papers, VIII mezhdunarodnaya konferentsiya “Sol'vatatsiya i kompleksoobrazovanie” (VIII Intern. Conf. “Solvation and Complexation”), Ivanovo, 2001, p. 16.
Fedorov, V.A., Doctoral (Chem.) Dissertation, Ivanovo, 1990.
Belevantsev, V.I. and Aseeva, I.D., Ross. Khim. Zh., 2000, no. 3, p. 58.
Fedorov, V.A., Isaev, I.D., and Eike, M.Yu., Koord. Khim., 1989, vol. 15, no. 9, p. 1152.
Kuz'mina, I.A. and Shormanov, V.A., Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 2000, vol. 43, no. 4, p. 71.
Rastvory neelektrolitov v zhidkosti (Solutions of Nonelectrolytes in Liquids), Nikiforov, M.Yu., Al'per, G.A., Durov, V.A. et al., Eds., Moscow: Nauka, 1989, p. 109.
Afanas'ev, V.N., Efremova, L.S., and Volkova, T.V., Fiziko-khimicheskie svoistva binarnykh rastvoritelei. Vodosoderzhashchie sistemy (Physicochemical Properties of Binary Solvents. Water-Containing Systems), Part 1, Ivanovo: Inst. Khim. Nevodn. Rastvorov Akad. Nauk SSSR, 1988.
Kukushkin, Yu.N., Khimiya koordinatsionnykh soedinenii (Chemistry of Coordination Compounds), Moscow: Vysshaya Shkola, 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fedorov, V.A., Robov, A.M. & Kirienko, M.A. Complexation in Mixed Nonaqueous Solvents. Russian Journal of Coordination Chemistry 29, 512–514 (2003). https://doi.org/10.1023/A:1024739315817
Issue Date:
DOI: https://doi.org/10.1023/A:1024739315817