Abstract
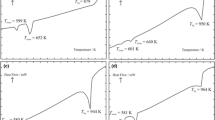
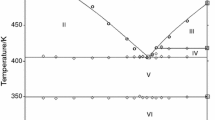
The heat capacity of solid NdBr3 was measured by Differential Scanning Calorimetry in the temperature range from 300 K up to the melting temperature. The heat capacity of liquid NdBr3 was also determined. These results were least-squares fitted to a temperature polynome. The melting enthalpy of NdBr3 was measured separately. DSC was used also to study phase equilibrium in the NdBr3-LiBr system. The results obtained provided a basis for constructing the phase diagram of the system under investigation. It represents a typical example of simple eutectic system. The eutectic composition, x(NdBr3)=0.278, was obtained from the Tamman construction. This eutectic mixture melts at 678 K. The electrical conductivity of NdBr3-LiBr liquid mixtures and of pure components was measured down to temperatures below solidification. Reflectance spectra of the pure components and their solid mixtures (after homogenisation in the liquid state) with different composition were recorded in order to confirm the reliability of the constructed phase diagram.
Similar content being viewed by others
References
M. Gaune-Escard, L. Rycerz, W. Szczepaniak and A. Bogacz, J. Alloys Comp., 204 (1994) 193.
M. Gaune-Escard, A. Bogacz, L. Rycerz and W. Szczepaniak, J. Alloys Comp., 235 (1996) 176.
L. Rycerz and M. Gaune-Escard, Z. Naturforsch., 57 a (2002) 79.
L. Rycerz and M. Gaune-Escard, Z. Naturforsch., 57 a (2002) 215.
L. Rycerz and M. Gaune-Escard, J. Therm. Anal. Cal., 56 (1999) 355.
M. Gaune-Escard and L. Rycerz, Z. Naturforsch., 54 a (1999) 229.
Y. Fouque, M. Gaune-Escard, W. Szczepaniak and A. Bogacz, J. Chim. Phys., 75 (1978) 360.
G. J. Janz, Materials Science Forum, 73–75 (1991) 707.
A. S. Dworkin and M. A. Bredig, High Temp. Sci., 31 (1971) 81.
I. Barin, O. Knacke and O. Kubaschewski, Thermochemical Properties of Inorganic Substances, Supplement, Springer-Verlag Berlin, Heidelberg, New York 1977.
A. S. Dworkin and M. A. Bredig, J. Phys. Chem., 67 (1963) 2499.
C. E. Wick and F. E. Block, Thermodynamic Properties of 65 Elements — Their Oxides, Halides, Carbides and Nitrides, Bureau of Mines, Bull. 605, US Gov. Printing Office, Washington 1963.
O. Kubaschewski, C. B. Alcoock and P. J. Spencer, Materials Thermochemistry, 6th Edition, Pergamon Press Ltd, 1993.
E. H. P. Cordfunke and R. J. M. Konings, Thermochim. Acta, 375 (2001) 17.
G. Vogel, Z. Anorg. Allg. Chem., 388 (1972) 43.
R. Blachnik and A. Jaeger-Kasper, Z. Anorg. Allg. Chem., 461 (1980) 74.
A. B. P. Lever, Inorganic Electronic Spectroscopy, 2nd Ed. Elsevier, Amsterdam, Oxford, New York, Tokyo 1984.
A. S. Dworkin, H. R. Bronstein and M. A. Bredig, J. Phys. Chem., 67 (1963) 2715.
G. Vogel, Diss. Clausthal T. K. 1970, p. 68.
G. J. Janz, NIST Standard Reference Database 27, Version 2.0 (1992).
I. S. Yaffe, E. R. Van Artsdalen ORNL — 2159 (1956) 77.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rycerz, L., Ingier-Stocka, E., Golonka-Cieślak, M. et al. Thermal and conductometric studies of NdBr3 and NdBr3-LiBr binary system. Journal of Thermal Analysis and Calorimetry 72, 241–251 (2003). https://doi.org/10.1023/A:1023940324328
Issue Date:
DOI: https://doi.org/10.1023/A:1023940324328