Abstract
Purpose. The aim of this study was to estimate the molecular state of salicylamide on the surface of mesoporous silicas and to investigate the dissolution behavior of salicylamide from the solid dispersion.
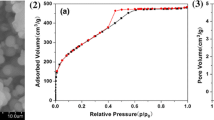
Methods. Folded sheets mesoporous material (FSM-16) were used as a porous material. The molecular state of salicylamide was estimated by powder X-ray diffractometry, infrared spectroscopy, and fluorescence spectroscopy.
Results. The molecular state of salicylamide can be changed by simple blending with FSM-16. When a physical mixture of 25% salicylamide and 75% FSM-16 was heated at 120°C for 3 h, amorphization of salicylamide was observed from the powder X-ray diffraction pattern. The fluorescence emission peak of salicylamide at 433.5 nm shifted to a longer wavelength of 447.5 nm after heating. Changes in fluorescence decay curve suggested that salicylamide molecules were dispersed into the hexagonal FSM-16 channels during the heating process. Enhanced dissolution in the initial stage of salicylamide from the sealed heated sample was observed in comparison with salicylamide crystals.
Conclusions. Heat treatment of a physical mixture of salicylamide with FSM-16 gave a solid dispersion in which the salicylamide molecules changed to an amorphous state by adsorption onto the FSM-16 channels. Amorphization of salicylamide contributed to the improvement of dissolution.
Similar content being viewed by others
References
S. Okonogi, T. Oguchi, E. Yonemochi, S. Puttipipatkhachon, and K. Yamamoto. Improved dissolution of ofloxacin via solid dispersion. Int. J. Pharm. 156:175–180 (1997).
T. Oguchi, Y. Tozuka, S. Okonogi, E. Yonemochi, and K. Yamamoto. Improved dissolution of naproxen from solid dispersions with porous materials. J. Pharm. Sci. Technol. Jpn. 57:168–173 (1997).
J. M. Kisler, A. Dähler, G. W. Stevens, and A. J. O'Connor. Separation of biological molecules using mesoporous molecular sieves. Micropor. Mesopor. Mater 44–45:769–774 (2001).
K. Matsumoto, Y. Nakai, E. Yonemochi, T. Oguchi, and K. Yamamoto. Aspirin hydrolysis in mixtures with porous crystalline cellulose. Drug Stability 1:93–97 (1996).
Y. Fukushima and S. Inagaki. Nano-scale structure control of mesoporous silica. Mater. Sci. Eng. A 217–218:116–118 (1996).
Y. Sakamoto, S. Inagaki, T. Ohsuna, N. Ohnishi, Y. Fukushima, Y. Nozue, and O. Terasaki. Structure analysis of mesoporous material ‘FSM-16’ Studies by electron microscopy and X-ray diffraction. Micropor. Mesopor. Mater. 21:589–596 (1998).
D. Shouro, Y. Moria, T. Nakajima, and S. Mishima. Mesoporous silica FSM-16 catalysts modified with various oxides for the vapor-phase beckmann rearrangement of cyclohexanone oxime. Appl. Catal. A: Gen. 198:275–282 (2000).
A. Fukuoka, N. Higashimoto, Y. Sakamoto, and S. Inagaki. Y.i Fukushima, and M Ichikawa. Preparation and catalysis of Pt and Rh nanowires and particles in FSM-16. Micropor. Mesopor. Mater 48:171–179 (2001).
T. Ishikawa, M. Matsuda, A. Yasukawa, K. Kandori, S. Inagaki, T. Fukushima, and S. Kondo. Surface silanol groups of mesoporous silica FSM-16. J. Chem. Soc. Faraday Trans. 92:1985–1990 (1996).
J. Palomar and J. L. G. De Paz. and J. Catalan. Vibrational study of intramolecular hydrogen bonding in o-hydroxybenzoil compounds. Chem. Phys. 246:167–208 (1999).
Y. Sasada and T. Takano, and M. Kakudo. Crystal structure of Salicylamide. Bull. Chem. Soc. Jpn. 37:940–946 (1964).
Y. Tozuka, E. Yonemochi, T. Oguchi, and K. Yamamoto. Molecular states of 2-naphthoic acid in solid disersions with porous crystalline cellulose, as investigated by fluorescence spectroscopy. Bull. Chem. Soc. Jpn. 73:1567–1572 (2000).
K. Kaneko. Molecular assembly formation in a solid nanospace. Colloids Surfaces A: Physicochem. Eng. Aspects 109:319–333 (1996).
T. Ohkubo, T. Iiyama, and K. Kaneko. Organized structures of methanol in carbon nanospaces at 303 K studies with in situ X-ray diffraction. Chem. Phys. Lett. 312:191–195 (1999).
T. Suzuki, R. Kobori, and K. Kaneko. Grand canonical Monte Carlo simulation-assisted pore-width determination of molecular sieve carbons by use of ambient temperature N2 adsorption. Carbon 38:630–633 (2000).
J. Miyawaki and K. Kaneko. Pore width dependence of the temperature change of the confined methane density in slit-shaped micropores. Chem. Phys. Lett. 337:243–247 (2001).
Y. Tozuka, E. Yonemochi, T. Oguchi, and K. Yamamoto. Fluorescence studies of pyrene adsorption on porous crystalline cellulose. J. Colloid Interface Sci. 205:510–515 (1998).
E. Yonemochi. M Kojinma, A. Nakatsuji, S. Okonogi, T. Oguchi, Y. Nakai, and K. Yamamoto. Thermal behavior of methyl p-hydroxybenzoate in controlled-pore glass solid dispersion. J. Colloid Interface Sci. 173:186–191 (1995).
A. Matsumoto, T. Sasaki, N. Nishijima, and K. Tsutsumi. Thermal stability and hydrophobicity of mesoporous silica FSM-16. Colloids and Surfaces A. Physicochem. Eng. Aspects 203:185–193 (2002).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tozuka, Y., Oguchi, T. & Yamamoto, K. Adsorption and Entrapment of Salicylamide Molecules into the Mesoporous Structure of Folded Sheets Mesoporous Material (FSM-16). Pharm Res 20, 926–930 (2003). https://doi.org/10.1023/A:1023851622676
Issue Date:
DOI: https://doi.org/10.1023/A:1023851622676