Abstract
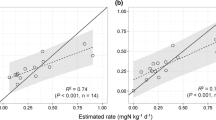
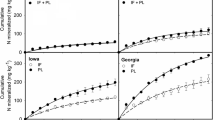
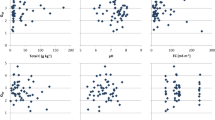
The N mineralized from soil organic matter provides an important portion of N available for crop production. The objective of this study was to determine the amount of spatial variability in N mineralization potential in a field and to evaluate three different methods that might be used to estimate this variability. The three methods tested included predicting the N mineralized from surface soil properties as well as from a biological and a chemical procedure. Three soils varying in N mineralization potential were selected for the study from a field in the Georgia Coastal Plain. The N mineralized from these soils was determined by an N balance of unfertilized and cropped plots. The amount of N mineralized could not be reliably predicted from surface soil organic C, although surface soil clay concentration was positively correlated with the N mineralized. The N mineralized that was predicted using mineralization parameters determined by aerobic incubation, adjusted daily for soil water content and temperature, was approximately 50% of the field measurements of N mineralized. The values of NH4-N extracted with hot 2 M KCl were related significantly to N mineralized in the field (r2= 0.60) and also to the zero order rate constant of mineralization, k0 (r2= 0.77), determined from the N mineralized in the aerobic laboratory incubation.
Similar content being viewed by others
References
Baker W.H. and Thompson T.L. 1992. Determination of total nitrogen in plant samples by Kjeldahl. In: Plank C.O. (ed.), Plant Analysis Reference Procedures for the Southern Region of the United States. Southern Cooperative Series Bulletin 368. The Georgia Agricultural Experiment Stations, University of Georgia, Athens, Georgia 71 pp.
Cabrera M.L. 1993. Modeling the flush of nitrogen mineralization caused by drying and rewetting soils. Soil Sci. Soc. Am. J. 57: 63-66.
Cabrera M.L. and Kissel D.E. 1988a. Evaluation of a method to predict nitrogen mineralization from soil organic matter under field conditions. Soil Sci. Soc. Am. J. 52: 1027-1031.
Cabrera M.L. and Kissel D.E. 1988b. Length of incubation time affects the parameter values of the double exponential model of nitrogen mineralization. Soil Sci. Soc. Am. J. 52: 1186-1187.
Cabrera M.L. and Kissel D.E. 1988c. Potentially mineralizable nitrogen in disturbed and undisturbed soil samples. Soil Sci. Soc. Am. J. 52: 1010-1015.
Cabrera M.L., Kissel D.E. and Vigil M.F. 1994. Potential nitrogen mineralization: Laboratory and field evaluation. In: Havlin J.L. and Jacobson J.S. (eds), Soil Testing: Prospects for Improving Nutrient Recommendations. SSSA Special Publication 40. Soil Science Society of America, Madison, Wisconsin, pp. 15-30.
Campbell C.A., Jame Y.D. and De Jong R 1988. Predicting net nitrogen mineralization over a growing season: Model verification. Can J. Soil. Sci. 68: 537-552.
Craswell E.T. and Waring S.A. 1972a. Effect of grinding on the decomposition of soil organic matter: I. The mineralization of organic nitrogen in relation to soil type. Soil Biol. Biochem. 4: 427-433.
Craswell E.T. and Waring S.A. 1972b. Effect of grinding on the decomposition of soil organic matter: II. Oxygen uptake and nitrogen mineralization in virgin and cultivated cracking clay soils. Soil Biol. Biochem. 4: 435-442.
Craswell E.T., Saffigna P.G. and Waring S.A. 1970. The mineralization of organic nitrogen in dry soil aggregates of different sizes. Plant Soil 33: 383-392.
Crooke W.M. and Simpson W.E. 1971. Determination of ammonium Kjeldahl digest of crops by an automated procedure. J. Sci. Food Agric. 22: 9-10.
Deans J.R., Molina J.A.E. and Clapp C.E. 1986. Models for predicting potentially mineralizable nitrogen and decomposition rate constants. Soil Sci. Soc. Am. J. 50: 323-326.
Edwards A.P. and Bremner J.M. 1967. Microaggregates in soils. J. Soil. Sci. 18: 64-73.
Ellert B.H. and Bettany J.R. 1988. Comparison of kinetic models for describing net sulfur and nitrogen mineralization. Soil Sci. Soc. Am. J. 52: 1692-1702.
Gianello C. and Bremner J.M. 1986a. A simple chemical method of assessing potentially available organic nitrogen in soil. Commun. Soil Sci. Plant Anal. 17: 195-214.
Gianello C. and Bremner J.M. 1986b. Comparison of chemical methods of assessing potentially available organic nitrogen in soil. Commun. Soil Sci. Plant Anal. 17: 215-236.
Herlihy M. 1979. Nitrogen mineralization in soils of varying texture, moisture and organic matter. Plant Soil 53: 255-267.
Hess T.H. and Schmidt S.K. 1994. Improved procedure for obtaining statistically valid parameter estimates from soil respiration data. Soil Biol. Biochem. 27: 1-7.
Hiura K., Hattori T. and Furusaka Ch. 1976. Bacteriological studies on the mineralization of organic nitrogen in paddy soils: I. Effects of mechanical disruption of soils on ammonification and bacterial number. Soil Sci. Plant Nutr. 22: 459-465.
Keeney D.R. and Nelson D.W. 1982. Nitrogen - inorganic forms. In: Page A.L. (ed.), Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties. 2nd edn. Agron. Monogr. 9. 2nd edn. American Society of Agronomy, Madison, Wisconsin, pp. 643-698.
Kilmer V.J. and Alexander L.T. 1949. Methods of making mechanical analysis of soils. Soil Sci. 68: 15-24.
Kirsten W. 1983. Organic Elemental Analysis: Ultramicro, Micro, and Trace Methods. Academic Press/Harcourt Brace Jovanovich, New York.
Kissel D.E., Ritchie J.T. and Richardson C.W. 1975. A stress day concept to improve nitrogen fertilizer utilization: dryland grain sorghum in the Texas Blackland Prairie. MP-1201, College Station, Texas Agricultural Experiment Station.
Magdoff F.R., Ross D. and Amadon J. 1984. A soil test for nitrogen availability to corn. Soil Sci. Soc. Am. J. 48: 1301-1304.
Neeteson J.J. 1990. Development of nitrogen fertilizer recom mendations for arable crops in the Netherlands in relation to nitrate leaching. Fert. Res. 26: 291-298.
Oien A. and Selmer-Olsen A.R. 1980. A laboratory method for evaluation of available nitrogen in soil. Acta Agric. Scand. 30: 149-156.
Reference Soil Test Methods for the Southern Region of the United States. September 1983. Southern Cooperative Series Bulletin 289, 40 pp. The Georgia Agricultural Experiment Stations, University of Georgia, Athens, Georgia.
Rovira A.D. and Greacen E.L. 1957. The effect of aggregate disruption on the activity of microorganisms in the soil. Austr. J. Agric. Res. 8: 659-673.
SAS Institute Inc. 1985. SAS User's Guide: Statistics. 6th edn. SAS Institute, Inc., Cary, North Carolina.
Schepers J.S. and Meisinger J.J. 1994. Field indicators of nitrogen mineralization. In: Havlin J.L. and Jacobson J.S. (eds), Soil Testing: Prospects for Improving Nutrient Recommendations. SSSA Special Publication 40. Soil Science Society of America, Madison, Wisconsin, pp. 31-47.
Stanford G. and Epstein E. 1974. Nitrogen mineralization-water relations in soils. Soil Sci. Soc. Am. Proc. 38: 103-107.
Stanford G. and Smith S.J. 1972. Nitrogen mineralization potentials of soils. Soil Sci. Soc. Am. Proc. 36: 465-472.
Stanford G., Frere M.H. and Schwaninger D.H. 1973. Temperature coefficient of soil nitrogen mineralization. Soil Sci. 115: 321-323.
Van Genuchten M.Th. 1980. A closed-form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci. Soc. Am. J. 44: 892-898.
Vigil M.F. and Kissel D.E. 1995. Rate of nitrogen mineralized from incorporated crop residues as influenced by temperature. Soil Sci. Soc. Am. J. 59: 1636-1644.
Wagenet R.J. and Hutson J.L., Water Resource Institute 1989. Leaching estimation and chemistry model: LEACHM. Cornell University, Ithaca, New York.
Waring S.A. and Bremner J.M. 1964. Effect of soil mesh-size on the estimation of mineralizable nitrogen in soils. Nature (London) 202: 1141-1141.
Whitehead D.C. 1981. An improved chemical extraction method for predicting the supply of available soil nitrogen. J. Sci. Food Agric. 32: 359-365.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Egelkraut, T., Kissel, D., Cabrera, M. et al. Predicting N mineralized in a Georgia Coastal Plain field. Nutrient Cycling in Agroecosystems 66, 1–12 (2003). https://doi.org/10.1023/A:1023306500473
Issue Date:
DOI: https://doi.org/10.1023/A:1023306500473