Abstract
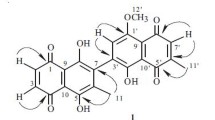
The title compound C20H24O4 crystallizes in the orthorhombic space group P21212, with a = 10.245(3), b = 17.103(3), c = 9.879(3) Å, D c = 1.260 g cm −3, and Z = 4. Nepetanudone is postulated to be formed by a [4+4] cycloaddition of two 5,9-dehydronepetalactone moieties to give an asymmetric dimer in which each pair of atoms is magnetically nonequivalent. The compound was recovered from extracts of Nepeta nuda ssp. albiflora.
Similar content being viewed by others
References
De Pooter, H.L.; Nicolai, B.; De Laet, J.; De Buyck, L.F.; Schamp, N.M.; Goetghebeur, P. Flavour Frag. J. 1988, 3, 155.
Davis, P.H. Flora of Turkey and the East Aegean Islands; Vol. 7; Edinburgh University Press: Edinburgh, 1982; pp. 264–282.
Kökdil, G.; Yalçin, S.M., Topcu, G. Turk. J. Chem. 1998 submitted.
Ahmad, V.U.; Bano, S. Tetrahedron Lett. 1981, 22, 1715.
Cotrim, H.C.; Barroso, J.G.; Figueiredo, A.C.; Pais, M.S.S.; Scheffer, J.J.C. Flavour Fragr. J. 1994, 9, 71.
Bourrel, C.; Perineau, F.; Michel, G. Bessiere, J.M. J. Essential Oil Res. 1993, 5, 159.
Urones, J.G.; Lithgow-Bertelloni, A.M.; Sexmero, M.J.; Marcos, I.S.; Basabe, P.; Moro, R.F. Anal. Quim. 1991, 87, 933.
Sheldrick, G.M. SHELXS86. Program for the Solution of Crystal Structures; University of Göttingen: Germany, 1986.
TEXSAN-TEXRAY Structure Analysis Package; Molecular Structure Corporation. Vers. 1.7-1. March 1995.
Spek, A.L. Acta Crystallogr. 1982, 29, 203.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kökdil, G., Topcu, G., Krawiec, M. et al. Nepetanudone, a dimer of the α-pyrone 5,9-dehydronepetalactone. Journal of Chemical Crystallography 28, 517–519 (1998). https://doi.org/10.1023/A:1023287804704
Issue Date:
DOI: https://doi.org/10.1023/A:1023287804704