Abstract
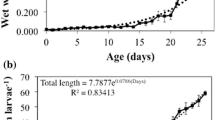
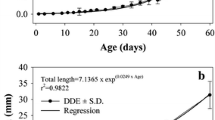
The temporal evolution of chymotrypsin activity during early ontogeny of laboratory reared red drum larvae was accomplished using a combination of biochemical assays and electrophoretic methods (substrate SDS-PAGE). Optimal functional conditions for chymotrypsin were also determined. Chymotrypsin activity was first detected prior to the onset of exogenous feeding. Total chymotrypsin activity increased with age and standard length. Specific activity was greatest on day 10 post-hatch. Maximal chymotrypsin activity was observed at 50 °C, pH 7.8, and Ca2+ concentration of 25 mM. Using substrate gel electrophoresis and specific inhibitors the molecular weight of red drum chymotrypsin was estimated to be 26–27 kD. Our results indicate that the digestive system of red drum larvae is capable of alkaline proteolysis before first feeding and suggest that chymotrypsin may have potential as an indicator of nutritional condition.
Similar content being viewed by others
REFERENCES
Alarcon, F.J., Diaz, M., Moyano, F.J. and Abellan, E. 1998. Characterization and functional properties of digestive proteases in two sparids; gilthead seabream (Sparus aurata) and common dentex (Dentex dentex). Fish Physiol. Biochem. 19: 257–267.
Applebaum, S.L. and Holt, G.J. (In press). The digestive protease, chymotrypsin, as an indicator of nutritional condition in larval red drum (Sciaenops ocellatus). Mar. Biol.
Arnold, C.R. 1988. Controlled year-round spawning of red drum (Sciaenops ocellatus) in captivity. Contrib. Mar. Sci. 30 (Suppl.): 65–70.
Ásgeirsson, B. and Bjarnason, J.B. 1991. Structural and kinetic properties of chymotrypsin from Atlantic cod (Gadus morhua). Comparison with bovine chymotrypsin. Comp. Biochem. Physiol. 99B: 327–335.
Baragi, V. and Lovell, R.T. 1986. Digestive enzyme activities in striped bass from first feeding through larval development. Trans. Am. Fish. Soc. 115: 478–484.
Clement, G.E. and Bender, M.L. 1963. The effect of aprotic dipolar organic solvents on the kinetics of alpha-chymotrypsin-catalysed hydrolysis. Biochemistry 2: 836–843.
Chong, A.S.C., Hashim, R., Chow-Yang, L. and Ali, A.B. 2002. Partial characterization and activities of proteases from the digestive tract of discus fish (Symphysodon aequifasciata). Aquaculture 203: 321–333.
Conceição, L.E.C., Houlihan, D.F. and Verreth, J.A.J. 1997. Fast growth, protein turnover and costs of protein metabolism in yolk-sac larvae of the African catfish (Clarias gariepinus). Fish Physiol. Biochem. 16: 291–302.
Dabrowski, K. 1984. The feeding of fish larvae – Present state of the art and perspectives. Reprod. Nutr. Dev. 24: 807–833.
Fuiman, L.A. 1994. The interplay of ontogeny and scaling in the interactions of fish larvae and their predators. J. Fish Biol. 45: 55–79.
Garcia-Carreno, F.L. and Haard, N.F. 1993. Substrate gel electrophoresis for composition and molecular weight of proteinase classes in langostilla (Pleuroncodes planipes) and crayfish (Pacifastacus astacus) extracts. J. Food Biochem. 17: 97–113.
Garcia-Ortega, A., Verreth, J. and Segner, H. 2000. Post-prandial protease activity in the digestive tract of African catfish Clarias gariepinus larvae fed decapsulated cysts of artemia. Fish Physiol. Biochem. 22: 237–244.
Gawlicka, A., Parent, B., Horn, M.H., Ross, N., Opstad, I. and Torrissen, O.J. 2000. Activity of digestive enzymes in yolk-sac larvae of Atlantic halibut (Hippoglossus hippoglossus): indication of readiness for first feeding. Aquaculture 184: 303–314.
Govoni, J.J., Boehlert, G.W. and Watanabe, Y. 1986. The physiology of digestion in fish larvae. Environ. Biol. Fish 16: 59–77.
Hjelmeland, K., Huse, I., Jørgensen, T., Molvik, G. and Raa, J. 1984. Trypsin and trypsinogen as indices of growth and survival potential of cod (Gadus morhua L.) larvae. In: The Propagation of cod, Gadus morhua, Part 1. Edited by E. Dahl, D.S. Danielssen, E. Moksness and P. Solemdal. Institute of Marine Research, Flødevigen Biological Station.
Holt, G.J. and Holt, S.A. 2000. Vertical distribution and the role of physical processes in the feeding dynamics of two larval sciaenids Sciaenops ocellatus and Cynoscion nebulosus. Mar. Ecol. Prog. Ser. 193: 181–190.
Holt, G.J. 1993. Feeding larval red drum on microparticulate diets in a closed recirculating water system. J. World Aquacult. Soc. 24: 225–230.
Hummel, B.C.W. 1959. A modified spectrophotometric determination of chymotrypsin, trypsin, and thrombin. Can. J. Biochem. Physiol. 37: 1393–1399.
Jonas, E., Ragyanszki, M., Olah, J. and Boross, L. 1983. Proteolytic digestive enzymes of carnivorous (Silurus glanis L.), herbivorous (Hypophthalmichthys molitrix Val.) and omnivorous (Cyprinus carpio L.) fishes. Aquaculture 30: 145–154.
Kawai, S. and Ikeda, S. 1973. Studies on digestive enzymes of fishes. Part 4. Development of the digestive enzymes of carp and black sea bream after hatching. Bull. Jap. Soc. Sci. Fish. 39: 877–881.
Kristjánsson, M.M. and Nielsen, H.H. 1992. Purification and characterization of two chymotrypsin-like proteases from the pyloric caeca of rainbow trout (Oncorhynchus mykiss). Comp. Phys. Biol. 101B: 247–253.
Krzyzosiak, J. and Daniel, R.M. 1997. Isolation and characterization of two chymotrypsins from Allocyttus niger (black oreo dory) viscera. N.Z.J. Mar. Freshwat. Res. 31: 497–504.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lauff, M. and Hofer, R. 1984. Proteolytic enzymes in fish development and the importance of dietary enzymes. Aquaculture 37: 335–346.
Lazo, J.P. 1999. Development of the digestive system in red drum (Sciaenops ocellatus) larvae. Doctoral Dissertation. University of Texas at Austin.
Lazo, J.P., Dinis, M.T., Holt, G.J., Faulk, C. and Arnold, C.R. 2000. Co-feeding microparticulate diets with algae: toward eliminating the need of zooplankton at first feeding in larval red drum (Sciaenops ocellatus). Aquaculture 188: 339–351.
Lazo, J.P., Holt, G. and Arnold, C. 2000a. Ontogeny of pancreatic enzymes in larval red drum Sciaenops ocellatus. Aquacult. Nutr. 6: 183–192.
Lazo, J.P., Mendoza, R., Holt, G. J., Aguilera, C. and Arnold, C.R. Characterization of digestive enzymes during larval development of red drum (Sciaenops ocellatus). (submitted).
Lazo, J.P., Romaire, R.P. and Reigh, R.C. 1998. Evaluation of three in vitro enzyme assays for estimating protein digestibility in the Pacific white shrimp Penaeus vannamei. J.World Aquacult. Soc. 29: 441–450.
Moyano, F.J., Diaz, M., Alarcon, F.J. and Sarasquete, M.C. 1996. Characterization of digestive enzyme activity during larval development of gilthead seabream (Sparus aurata). Fish Physiol. Biochem. 15: 121–130.
Munilla-Moran, R., Stark, J.R. and Barbour, A. 1990. The role of exogenous enzymes in cultured turbot larvae (Scopthalmus maximus L.). Aquaculture 88: 337–350.
Oozeki, Y. and Bailey, K.M. 1995. Ontogenetic development of digestive enzyme activities in larval walleye pollock, Theragra chalcogramma. Mar. Biol. 122: 177–186.
Pedersen, B.H. 1993. Protein digestion in herring (Clupea harengus) larvae: Trypsinogen secretion, and effects of a transitory food restriction on mortality, growth and digestive enzyme con-tent. In: Physiological and Biochemical Aspects of Fish Development. Edited by B.T. Walther and H.J. Fhyn. University of Bergen, Norway.
Pedersen, B.H., Nilssen, E.M. and Hjelmeland, K. 1987. Variations in the content of trypsin and trypsinogen in larval herring (Clupea harengus) digesting copepod nauplii. Mar. Biol. 94: 171–181.
Ramakrishna, M., Hultin, H.O. and Racicot, W.F. 1987. Some kinetic properties of dogfish chymotrypsin. Comp. Phys. Biol. 87B: 25–30.
Ribeiro, L., Zambonino-Infante, J.L., Cahu, C. and Dinis, M.T. 1999. Development of digestive enzymes in larvae of Solea senegalensis, Kaup 1858. Aquaculture 179: 465–473.
Segner, H., Roesch, R., Schmidt, H. and von Poeppinghausen, K.J. 1989. Digestive enzymes in larval Coregonus lavaretus L. J. Fish Biol. 35: 249–263.
Smith, P.K., Krohn, R.I., Hermanson, G.T., Mallia, A.K. Gartner, F.H., Provenzano, M.D., Fujimoto, E.K., Goeke, M.M.B.J. and Klenk, D.C. 1985. Measurement of protein using bicichoninic acid. Anal. Biochem. 150: 76–85.
Stryer, L. 1988. Biochemistry. W.H. Freeman and Company, New York.
Tanaka, M. 1972. Studies on the structure and function of the digestive system in teleost larvae. Part 5: Epithelial changes in the posterior gut and protein digestion. Jap. J. Ichthyol. 19: 172–180.
Ueberschär, B. 1993. Measurement of proteolytic enzyme activity: Significance and application in larval fish research. In: Physiological and Biochemical Aspects of Fish Development. Edited by B.T. Walther an H.J. Fhyn. University of Bergen, Norway.
Ueberschär, B. 1995. The use of tryptic enzyme activity measurement as a nutritional condition index: Laboratory calibration data and field application. ICES Marine Science Symposium 201: 119–129.
Ueberschär, B. and Clemmesen, C. 1992. A comparison of the nutritional condition of herring larvae as determined by two biochemical methods-tryptic enzyme activity and RNA/DNA ratio measurements. ICES J. Mar. Sci. 49: 245–249.
Walford, J. and Lam, T.J. 1993. Development of digestive tract and proteolytic enzyme activity in seabass (Lates calcarifer) larvae and juveniles. Aquaculture 109: 187–205.
Wilcox, P.E. 1970. Chymotrypsinogens-chymotrypsins. In: Methods in Enzymology. Edited by G.E. Perlmann and L. Lorand. 19: 64–108.
Zambonino-Infante, J.L. and Cahu, C. 1994. Development and response to a diet change of some digestive enzymes in sea bass (Dicentrarchus labrax) larvae. Fish Physiol. Biochem. 12: 399–408.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Applebaum, S., Perez, R., Lazo, J. et al. Characterization of chymotrypsin activity during early ontogeny of larval red drum (Sciaenops ocellatus). Fish Physiology and Biochemistry 25, 291–300 (2001). https://doi.org/10.1023/A:1023202219919
Issue Date:
DOI: https://doi.org/10.1023/A:1023202219919