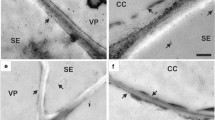
Abstract
Carbon assimilation in maize follows the C4 mechanism. This requires the tissue-specific and light-induced expression of a set of different genes involved in CO2 fixation as well as adaptations in the leaf anatomy including a reduced distance between vascular bundles compared to C3 plants. However, several maize tissues exist with larger bundle distances and there is significant evidence that CO2 fixation follows the C3 mechanism in these tissues. We isolated maize C3 and C4 tissues and quantified the accumulation of mRNAs encoding PEPC, ME, the small subunit of Rubisco, and PPDK. For this, primer systems for the specific and sensitive detection by real-time PCR were established. The observed patterns show the expected distribution for foliar leaf tissues. Also in total husk leaves, all transcripts under investigation were detected, albeit at a lower level. When mesophyll cells which are located distant from bundles were isolated from husk leaves, only accumulation of RbcS was observed. Comparing the expression of two genes encoding for isoenzymes of the small subunit of RbcS in the different tissues differential patterns of relative transcript abundance were observed. Transcripts for the DOF1 transcription factor involved in the activation of photosynthetic genes in maize were found in leaf tissues performing both C4 and C3 photosynthesis with highest accumulation levels in C4 mesophyll cells, whereas the homologous DOF2 gene was not expressed in any of the investigated samples. The results provide novel insights into the regulation of C3 and C4 carbon fixation pathways in maize.
Similar content being viewed by others
References
Al-Taher A, Bashein A, Nolan T, Hollingsworth M and Brady G (2000) Global cDNA amplification combined with real-time RT-PCR: accurate quantification of multiple human potassium channel genes at the single cell level. Yeast 17: 201–210
Antonielli M and Venanzi G (1979) Structural properties of the rachis and hypsophyll of the maize ear. Plant Sci Lett 15: 301–304
Chomczynski P (1993) A reagent for the single-step simultaneous isolation of RNA, DNA and proteins from cell and tissue samples. Biotechniques 15: 532–534
Crespo HM, Frean M, Cresswell CF and Tew J (1979) The occurrence of both C3 and C4 photosynthetic characteristics in a single Zea mays plant. Planta 147: 257–263
Cribb L, Hall LN and Langdale JA (2001) Four mutant alleles elucidate the role of the G2 protein in the development of C4 and C3 photosynthesizing maize tissues. Genetics 159: 787–797
Dengler NG and Nelson T (1999) Leaf structure and development in C4 Plants. In: Sage RF and Monson RK (eds) C4 Plant Biology, pp 133–172. Academic Press, San Diego
Dong L-Y, Masuda T, Kawamura T, Hata S and Izui K (1998) Cloning, expression, and characterization of a root-form phosphoenolpyruvate carboxylase from Zea mays: comparison with the C4-form enzyme. Plant Cell Physiol 39: 865–873
Ewing RM, Jenkins GI and Langdale JA (1998) Transcripts of maize RbcS genes accumulate differentially in C3 and C4 tissues. Plant Mol Biol 36: 593–599
Furumoto T, Hata S and Izui K (2000) Isolation and characterization of cDNAs for differentially accumulated transcripts between mesophyll cells and bundle sheath strands of maize leaves. Plant Cell Physiol 41: 1200–1209
Hall LN, Rossini L, Cribb L and Langdale JA (1998) GOLDEN2: a novel transcriptional regulator of cellular differentiation in the maize leaf. Plant Cell 10: 925–936
Heid CA, Stevens J, Livak KJ and Williams PM (1996) Real time quantitative PCR. Genome Res 6: 986–994.
Kanai R and Edwards GE (1999) The Biochemistry of C4 Photosynthesis. In: Sage RF and Monson RK (eds) C4 Plant Biology, pp 49–87. Academic Press, San Diego, California
Ku MSB, Kano Murakami Y and Matsuoka M (1996) Evolution and expression of C4 photosynthesis genes. Plant Physiol 111: 949–957
Lai LB, Tausta SL and Nelson TM (2002a) Differential regulation of transcripts encoding cytosolic NADP-malic enzyme in C3 and C4 Flaveria species. Plant Physiol 128: 140–149
Lai LB, Wang L and Nelson TM (2002b) Distinct but conserved functions for two chloroplastic NADP-malic enzyme isoforms in C3 and C4 Flaveria species. Plant Physiol 128: 125–139
Langdale JA, Zelitch I, Miller E and Nelson T (1988) Cell position and light influence C4 versus C3 patterns of photosynthetic gene expression in maize. EMBO J 7: 3643–3652
Nelson T and Langdale JA (1992a) Developmental genetics of C4photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 43: 25–47
Nelson T and Langdale J (1992b) Patterns of leaf development in C4 plants. Plant Cell 1: 3–13
Raeymaekers L (2000) Basic principles of quantitative PCR. Mol Biotechnol 15: 115–122
Ririe KM, Rasmussen RP and Wittwer CT (1997) Product differentiation by analysis of DNA melting curves during the polymerase chain reaction. Anal Biochem 245: 154–160
Rothermel BA and Nelson T (1989) Primary structure of the maize NADP-dependent malic enzyme. J Biol Chem 264: 19587– 19592
Schäffner AR and Sheen J (1992) Maize C4 photosynthesis involves differential regulation of phosphoenolpyruvate carboxylase genes. Plant J 2: 221–232
Sheen J (1991) Molecular mechanisms underlying the differential expression of maize pyruvate orthophosphate dikinase genes. Plant Cell 3: 225–246
Sheen J (1999) C4 gene expression. Annu Rev Plant Physiol Plant Mol Biol 50: 187–217
Sheen J and Bogorad L (1986) Expression of the ribulose-1,5-bisphosphate carboxylase large subunit gene and three small subunit genes in two cell types of maize leaves. EMBO J 5: 3417–3422
Sheen JY and Bogorad L (1987) Differential expression of C4 pathway genes in mesophyll and bundle sheath cells of greening maize leaves. J Biol Chem 262: 11726–11730
Wittwer CT, Ririe KM, Andrew RV, David DA, Gundry RA and Balis UJ (1997) The LightCycler: a microvolume multisample fluorimeter with rapid temperature control. Biotechniques 22: 176–181
Yakir D, Osmond B and Giles L (1991) Autotrophy in maize husk leaves: evaluation using natural abundance of stable isotopes. Plant Physiol 97: 1196–1198
Yanagisawa S (2000) Dof1 and Dof2 transcription factors are associated with expression of multiple genes involved in carbon metabolism in maize. Plant J 21: 281–288
Yanagisawa S and Sheen J (1998) Involvement of maize Dof zinc finger proteins in tissue-specific and light-regulated gene expression. Plant Cell 10: 75–89
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hahnen, S., Joeris, T., Kreuzaler, F. et al. Quantification of photosynthetic gene expression in maize C3 and C4 tissues by real-time PCR. Photosynthesis Research 75, 183–192 (2003). https://doi.org/10.1023/A:1022856715409
Issue Date:
DOI: https://doi.org/10.1023/A:1022856715409