Abstract
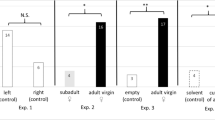
Male Loligo pealeii engage in frequent agonistic bouts to gain access to female mates while aggregated at communal egg beds. Male squids are attracted to eggs in the field and in the laboratory. It was recently demonstrated that visual detection followed by physical contact with egg capsules elicited male–male aggression. We tested specific physical and chemical features of the egg capsules that may cause this strong behavioral reaction. Male squids were presented with either natural or artificial egg stimuli and scored for four selected behaviors (egg touch, egg blowing, forward-lunge grab, and fin-beating), the last two of which are highly aggressive behaviors. First, squids were presented with natural eggs versus eggs sealed in agarose-coated tubes (ESACT), which eliminated both tactile and chemical stimuli. Second, males were presented with natural eggs versus eggs sealed in agarose coated tubes containing C18 Sep-Pak-purified extracts (TCPE) from squid egg capsules, which provided chemical cues from natural eggs without the physical stimulus of the egg capsules. Third, natural eggs versus heat-denatured eggs were tested to determine whether the active factor in natural eggs is heat-labile. Squids responded aggressively when contacting natural eggs and TCPE, whereas squids did not respond after touching ESACT or denatured eggs. These results suggest that aggressive behavior is elicited by a heat-labile factor that is embedded within squid egg capsules. This chemosensory cue appears to be a contact pheromone that stimulates the agonistic interactions that characterize the mating behavior of migratory squids on inshore spawning grounds.
Similar content being viewed by others
References
Adamo, S. A. and Hanlon, R. T. 1996. Do cuttlefish (Cephalopoda) signal their intentions to conspecifics during agonistic encounters? Anim. Behav. 52:73–81.
Archer, J. 1988. The Behavioural Biology of Aggression. Cambridge University Press, Cambridge.
Arnold, J. M. 1962. Mating behavior and social structure in Loligo pealei. Biol. Bull. 123:53–57.
Begnoche, V. L., Moore, S. K., Blum, N., vanGils, C., and Mayeri, E. 1996. Egg masses are a sign stimulus for release of bag cell peptides and reproductive behavior in Aplysia. J. Neurophysiol. 75:2162–2166.
Berrill, M. and Arsenault, M. 1984. The breeding behavior of a northern temperate Orconectid crayfish, Orconectes rusticus. Anim. Behav. 32:333–339.
Black, G. A. P., Rowell, T. W., and Dawe, E. G. 1987. Atlas of the biology and distribution of the squids Ilex illecebrocus and Loligo pealei in the northwest Atlantic. Can. Spec. Publ. of Fish. Aquat. Sci. 100:2–9.
Boal, J. G. 1997. Female choice of males in cuttlefish (Mollusca: Cephalopoda). Behaviour 134:975–988.
DiMarco, F. P. and Hanlon, R. T. 1997. Agonistic behavior in the squid Loligo plei (Loliginidae, Teuthoidea): fighting tactics and the effects of size and resource value. Ethology 103:89–108.
Emery, D. G. 1975. Ciliated sensory neurons in the lip of the squid Lolliguncula brevis Blaineville. Cell Tissue Res. 157:323–329.
Gilly, W. F. and Lucero, M. T. 1992. Behavioral responses to chemical stimulation of the olfactory organ in the squid Loligo opalescens. J. Exp. Biol. 162:209–229.
Hanlon, R. T. 1996. Evolutionary games that squids play: Fighting, courting, sneaking, and mating behaviors used for sexual selection in Loligo pealei. Biol.Bull. 191:309–310.
Hanlon, R. T. 1998. Mating systems and sexual selection in the squid Loligo: how might commercial fishing on spawning squids affect them? CalCOFI Rep. 39:92–100.
Hanlon, R. T. and Messenger, J. B. 1996. Cephalopod Behaviour. Cambridge University Press, Cambridge, UK.
Hirschenhauser, K., Moestl, E., Wallner, B., Dittami, J., and Kotrschal, K. 2000. Endocrine and behavioural responses of male greylag geese (Anser anser) to pairbond challenges during the reproductive season. Ethology 106:63–77.
Huntingford, F. A. and Turner, A. K. 1987. Animal Conflict. Chapman and Hall, New York.
Joanidopoulos, K. D. and Marwan, W. 1999. A combination of chemosensory and mechanosensory stimuli triggers the male mating response in the giant rotifer Asplanchna sieboldi. Ethology 105:465–475.
Johnsen, T. S., Zuk, M., and Fessler, E. A. 2001. Social dominance, male behaviour and mating in mixed-sex flocks of red jungle fowl. Behaviour 138:1–18.
Kemp, D. J. and Wiklund, C. 2001. Fighting without weaponry: a review of male-male contest competition in butterflies. Behav. Ecol. Sociobiol. 49:429–442.
King, A. J., Adamo, S. A., Hanlon, R. T. (in press) Agonsitic behaviour between male squid: squid egg mops provide sensory cues for swift conflict escalation. Anim. Behav.
Lonsdale, D. J., Frey, M. A., and Snell, T. W. 1998. The role of chemical signals in copepod reproduction. J. Mar. Syst. 15:1–12.
Painter, S. D., Clough, B., Garden, R. W., Sweedler, J. V., and Nagle, G. T. 1998. Characterization of Aplysia attractin, the first water-borne peptide pheromone in invertebrates. Biol. Bull. 194:120–131.
Painter, S. D., Clough, B., Akalal, D. B. G., and Nagle, G. 1999. Attractin, a water-bourne peptide pheremone in Aplysia. Invert. Reprod. Dev. 36:191–194.
Sokal, R. R. and Rohlf, F. J. 1995. Biometry: The Principles and Practice of Statistics in Biological Research, 3rd ed. W.H. Freeman and Company, New York.
Summers, W. C. 1983. Loligo pealei, pp. 115–142, in P. R. Boyle (ed.). Cephalopod Life Cycles, Vol. I: Species Accounts. Academic Press, London.
Walsh, K. 1970. Trypsinogens and trypsins of various species. Methods Enzymol. 19:41.
Woodhams, P. L. and Messenger, J. B. 1974. A note on the ultrastructure of the octopus olfactory organ. Cell Tissue Res. 152:253–258.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buresch, K.C., Boal, J.G., Knowles, J. et al. Contact Chemosensory Cues in Egg Bundles Elicit Male–Male Agonistic Conflicts in the Squid Loligo pealeii . J Chem Ecol 29, 547–560 (2003). https://doi.org/10.1023/A:1022846603591
Issue Date:
DOI: https://doi.org/10.1023/A:1022846603591