Abstract
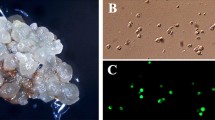
Culture conditions are described for sustained cell division and plant regeneration from protoplasts of rose (Rosa hybrida L. `Sumpath'). Protoplasts were enzymatically isolated from 2-week-old embryogenic cell suspension cultures. Freshly isolated protoplasts were plated as a thin layer onto protoplast culture medium (half-strength 21 Murashige and Skoog's medium containing 60 g l−1 myo-inositol, 4.4 μM BA, and 1.4 μM 2,4-D) at a density of 5×104 protoplasts ml−1. The plating efficiency reached 3.9% after 2 weeks of culture. However, few protoplasts underwent cell division when cultured in protoplast culture medium in which 60 g l−1 myo-inositol was replaced with the same osmolarity of 90 g l−1 mannitol, indicating that myo-inositol is essential for sustained cell division of protoplasts. Colonies were formed after 8 weeks of culture at a frequency of 0.2%. Colonies were then transferred to colony culture medium (0.4% Gelrite-solidified protoplast culture medium) and maintained by subculturing at 4-week intervals to form embryogenic calluses. Upon transfer to half-strength MS basal medium, embryogenic calluses gave rise to numerous somatic embryos. Somatic embryos were transferred to half-strength MS basal medium containing 48 mg l−1 ferric ethylenediamine di-(o-hydroxyphenylacetate), where they subsequently developed into plantlets at a frequency of 30.9%. The plantlets had the same chromosome number of 2n=3x=21 as the source plant. They were successfully transplanted to potting soil and grown to maturity in a greenhouse.
Similar content being viewed by others
References
Arya S, Liu JR & Eriksson T (1991) Plant regeneration from protoplasts of Panax ginseng (CA Meyer) through somatic embryogenesis. Plant Cell Rep. 10: 277-281
Darlington CD & Wylie AP (1956) Chromosome Atlas of Flower-ing Plants. In: Chromosome Atlas of Flowering Plants (pp 134- 138). Allen and Unwin, London
Frearson EM, Power JB & Cocking EC (1973) The isolation, culture and regeneration of Petunia leaf protoplasts. Dev. Biol. 33: 130-137
Gupta HS & Pattanayak A (1993) Plant regeneration from meso-phyll protoplasts of rice (Oryza sativa L.). Bio/Technology 11: 90-94
Kim SW, Oh SC & Liu JR (2002) Control of direct and indirect somatic embryogenesis in immature zygotic embryo cultures of rose (Rosa hybrida L.) by exogenous growth regulators. Plant Cell Tiss. Org. Cult. (submitted)
Krishnamurthy KV, Hendre RR, Godbole DA, Kulkarni VM, Mascarenhas AF & Jagannathan V(1979) Isolation and regeneration of rose bud callus protoplasts (Rosa sp. cv. Soraya). Plant Sci. Lett. 15: 135-137
Marchant R, Davey MR & Power JB (1997) Isolation and culture of mesophyll protoplasts from Rosa hybrida. Plant Cell Tiss. Org. Cult. 50: 131-134
Matthews D, Mottley J, Horan I & Roberts AV(1991) A protoplast to plant system in roses. Plant Cell Tiss. Org. Cult. 24: 173-180
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15: 473-497
Pearce RS & Cocking EC (1973) Behavior in culture of isolated protoplasts from 'Paul's Scarlet' rose suspension culture cells. Protoplasma 77: 165-180
Shillito RD, Paszkowski J & Potrykus I (1983) Agarose plating and a bead type culture technique enable and stimulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep. 2: 244-247
Strauss A & Potrykus I (1980) Callus formation from protoplasts of cell suspension cultures of Rosa 'Paul's Scarlet'. Physiol. Plant. 48: 15-20
Van der Salm TPM, Van der Toorn CJG, Hanisch ten Cate CH, Dubois LAM, De Vries DP & Dons HJM (1994) Importance of the iron chelate formula for micropropagation of Rosa hybrida L. 'Money'. Plant Cell Tiss. Org. Cult. 37: 73-77
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S.W., Oh, S.C., In, D.S. et al. Plant regeneration of rose (Rosa hybridia) from embryogenic cell-derived protoplasts. Plant Cell, Tissue and Organ Culture 73, 15–19 (2003). https://doi.org/10.1023/A:1022693605436
Issue Date:
DOI: https://doi.org/10.1023/A:1022693605436