Abstract
Future success using chemotherapy against human gliomas may result from exploiting unique molecular vulnerabilities of these tumors. Chemotherapy frequently results in DNA damage. When such damage is sensed by the cell, programmed cell death, or apoptosis, may be initiated. However, chemotherapy-induced DNA damage may activate nuclear factor kappa B (NF-κB) and block apoptosis. We inhibited NF-κB using a gene therapy approach to determine whether this would render human glioma cells more susceptible to chemotherapy. U87 and U251 glioma cell lines were infected with either treatment adenovirus containing the gene for a mutant non-degradable form of IκBα, which is an inhibitor of NF-κB nuclear translocation, or empty control virus. Following viral infection, cells were treated either with BCNU, carboplatin, tumor necrosis factor alpha (TNF-α), or SN-38.
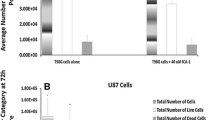
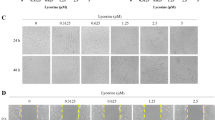
Chemotherapy resulted in a marked increase in active intranuclear NF-κB. This response was greatly decreased by insertion of the mutant repressor gene. Similarly, a significant increase in cell killing by all chemotherapy age was demonstrated following infection with treatment virus. Expression of the mutant repressor gene also resulted in increased apoptosis by TUNEL assay following chemotherapy.
Numerous genes are responsible for glioma chemoresistance. DNA damage by chemotherapy may induce the antiapoptotic factor NF-κB and prevent programmed cell death. Insertion of a mutant inhibitor of NF-κB strips cells of this antiapoptotic defense and renders them more susceptible to killing by chemotherapy via increased apoptosis.
Similar content being viewed by others
References
Beg AA, Baldwin Jr AS: Activation of multiple NF-KB/Rel DNA-binding complexes by tumor necrosis factor. Oncogene 9: 1487–1492, 1994
Mercurio F, Manning AM: Multiple signals converging on NF-KB. Curr Opin Cell Biol 11: 226–232, 1999
Beg A, Baldwin Jr AS: The IKB proteins: multifunctional regulators of Rel/NF-KB transcription factors. Genes Dev 7: 2064–2070, 1993
Ouaaz F, Li M, Beg AA: A critical role for the RelA subunit of nuclear factor KB in regulation of multiple immuneresponse genes and in fas-induced cell death. J Exp Med 189: 999–1004, 1999
Webster GA, Perkins ND: Transcriptional cross talk between NF-KB and p53. Mol Cell Biol 19: 3485–3495, 1999
Chen Z, Hagler J, Palombella VJ, Melandri F, Scherer D, Ballard D, Maniatis T: Signal-induced site-specific phosphorylation targets IKB? to the ubiquitin-proteasome pathway. Genes Dev 9: 1586–1597, 1995
DiDonato J, Mercurio F, Rosette C, Wu-Li J, Suyang H, Ghosh S, Karin M: Mapping of the inducible IKB phosphorylation sites that signal its ubiquination and degradation. Mol Cell Biol 16: 1295–1304, 1996
Yaron A, Gonen H, Hatzubai A, Jung S, Beyth S, Mercurio F, Manning AM, Ciechanover A, Ben-Neriah Y: Inhibition of NF-KB cellular function via specific targeting of the I?B ubiquitin ligase. EMBO J 16: 101–107, 1997
Ghosh S, May MJ, Kopp EB: NF-KB and rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol 16: 225–260, 1998
Reuther JY, Reuther GW, Cortez D, Pendergast AM, Baldwin Jr AS: A requirement for NF-K;B activation in Bcr-Abl-mediated transformation. Gene Dev 12: 968–981, 1998
Tamatanti M, Che YH, Matsuzaki H, Ogawa S, Okado H, Miyake S, Mizuno T, Tohyama M: Tumor necrosis factor 196 induces bcl-2 and bcl-x expression through NF-KB activation in primary hippocampal neurons. J Biol Chem 274: 8531–8538, 1999
Pajonk F, Pajonk K, McBride WH: Inhibition of NF-KB, clonogenicity and radiosensitivity of human cancer cells. J Natl Cancer Inst 91: 1956–1960, 1999
Wang C, Mayo MW, Baldwin Jr AS: TNF-? and cancer therapy-induced apoptosis: potentiation by inhibition of NF-KB. Science 274: 784–787, 1996
Cusack Jr JC, Liu R, Baldwin Jr AS: Inducible chemoresistance to 7-ethyl-10-[4-(1-piperidini)-1-piperidino]-carbonyloxycamptothecin (CPT-11) in colorectal cancer cells and a xenograft model is overcome by inhibition of nuclear factor-KB activation. Cancer Res 60: 2323–2330, 2000
Wang C, Cusack Jr JC, Liu R, Baldwin Jr AS: Control of inducible chemoresistance: enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-KB. Nat Med 5: 412–417, 1999
Russo SM, Tepper JE, Baldwin Jr AS, Liu R, Adams J, Elliott P, Cusack Jr JC: Enhancement of radiosensitivity by proteasome inhibition: implications for a role of NF-KB. Int J Radiation Oncology Biol Phys 50(1): 183–193, 2001
Shinoura N, Yamamoto N, Yoshida Y, Fujita T, Saito N, Asai A, Kirino T, Hamada H: Adenovirus-mediated gene transduction of IB or IKB Plus bax gene drastically enhances tumor necrosis factor (TNF)-induced apoptosis in human gliomas. Jpn J Cancer Res 91: 41–51, 2000
Miyakoshi J, Yagi K: Inhibition of IKB? phosphorylation at serine and tyrosine acts independently on sensitization to DNA damaging agents in human glioma cells. Br J Cancer 82(1): 28–33, 2000
Van Antwerp DJ, Martin SJ, Kafri S, Green DR, Verma IM: Suppression of TNF-?-induced apoptosis by NF-KB. Science 274: 787–789, 1996
Otsuka G, Nagaya T, Saito K, Mizuno M, Yoshida J, Seo H: Inhibition of nuclear factor-KB activation confers sensitivity to tumor necrosis factor-? by impairment of cell cycle progression in human glioma cells. Cancer Res 59: 4446–4452, 1999
Mayo MW, Wang C, Cogswell PC, Rogers-Graham KS, Der CJ, Baldwin Jr AS: Requirement of NF-KB activation to suppress p53-independent apoptosis induced by oncogenic ras. Science 278: 1812–1815, 1997
Schrum LW, Black D, Iimuro Y, Rippe RA, Brenner DA, Behrns K: C-jun does not mediate hepatocyte apoptosis following NF-KB inhibition and partial hepatectomy. J Surg Res 88: 142–149, 2000
O'Neill LAJ, Kaltschmidt C: NF-KB: a crucial transcription factor for glial and neuronal cell function. Trends Neurosci 20: 252–258, 1997
Moerman AM., Mao L, Lucas MM, Barger SW: Characterization of a neuronal KB-binding factor distinct form NF-KB. Mol Brain Res 67: 303–315,1999
Baldwin AS: The NF-KB and IKB proteins: new discoveries and insights. Annu Rev Immunol 14: 649–681, 1996
Bushdid P, Brantley D, Yull F, Blaeuer G, Hoffman L, Niswander L, Kerr L: Inhibition of NF-KB in disruption of the apical ectodermal ridges and aberrant limb morphogenesis. Nature 392: 615–619, 1998
Franzoso G, Carlson L, Xing L, Poljak L, Shores E, Brown K, Leonardi A, Tram T, Boyce B, Siebenlist U: Requirements for NF-KB in osteoclast and B-cell development.Genes Dev 11: 3482–3496, 1997
Iotsova V, Caamano J, Loy J, Yang Y, Lewin A, Bravo R: Osteopetrosis in mice lacking NF-KB1 and NF-?B2. Nat Med 3: 1285–1289, 1997
Deryckere F, Burger HG: Tumor necrosis factor ? induces the adenovirus early 3 promoter by activation of NF-KB. J Biol Chem 271: 30249–30255, 1996
Limbourg FP, Stadtler H, Chinnadurai G, Baeurle PA, Schmitz ML: A hydrophobic region within the adenovirus E1b 19 kDa protein is necessary for the transient inhibition of NF-KB activated by different stimuli. J Biol Chem 271: 20392–20398, 1996
Schmitz ML, Indorf A, Limbourg FP, Stadtler H, Traenckner HB-M, Baeuerle PA: The dual effect of adenovirus type 5 E1A 13S protein on NF-KB activation is antagonized by E1B 19K. Mol Cell. Biol 15: 4052–4063, 1996
Shao R, Karunagaran D, Zhou BP, Li K, Lo S, Deng J, Chiao P, Hung M: Inhibition of nuclear factor-KB activity is involved in E1A-mediated sensitization of radiationinduced apoptosis. J Biol Chem 272: 32739–32742, 1997
Lieber A, He C, Meuse L, Himeda C, Wilson C, Kay MA: Inhibition of NF-KB Activation in combination with bcl-2 expression allows for persistence of first-generation adenovirus vectors in the mouse liver. J Virol 72:9267–9277, 1998
Viola JJ, Ram Z, Walbridge S, Oshiro Em, Trapnell B, Tao-Cheng JH, Oldfield EH: Adenovirally mediated gene transfer into experimental solid brain tumors and leptomeningeal cancer cells. J Neurosurg 82: 70–76, 1995
Peltekian E, Parrish E, Bouchard C, Peschanski M, Lisovoski F: Adenovirus-mediated gene transfer to the brain: methodological assessment. J Neurosci Methods 71: 77–84, 1997
Lang FF, Yung WKA, Sawaya R, Tofilon PJ: Adenovirusmediated p53 gene therapy for human gliomas. Neurosurg 45(5): 1093–1104, 1999
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weaver, K.D., Yeyeodu, S., Cusack, J.C. et al. Potentiation of Chemotherapeutic Agents following Antagonism of Nuclear Factor kappa B in Human Gliomas. J Neurooncol 61, 187–196 (2003). https://doi.org/10.1023/A:1022554824129
Issue Date:
DOI: https://doi.org/10.1023/A:1022554824129