Abstract
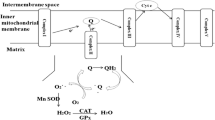
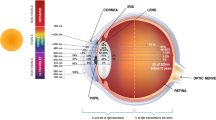
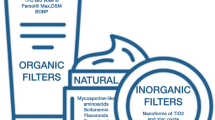
The purpose of the present study was to determine in vitro the effect of sodium azide, ethanol, trans-β-carotene, and the reduced form of glutathione on phototransformations in the lens crystallin. These photostabilizers show a specific affinity for different kinds of free radicals. The water-soluble protein from the cortical part of the eye was irradiated with doses of UV C ranging from 0 to 4.07 J/cm2. Changes in the structure of the crystallins have been monitored by steady-state absorption and fluorescence spectroscopy. Irradiation of dialyzed samples of these proteins at a wavelength of 254 nm (1.13 ± 0.02 mW/cm2) caused photooxidation of aromatic residues; the crystallin solutions became opaque and turbid. The samples displayed increasing attenuance at a wavelength of 280 nm as photodamage proceeded. The fluorescence of tryptophan at 333 nm systematically decreased and a new band between 400 and 500 nm appeared during the UV C irradiation. Our results show that the antioxidants can protect proteins from UV C-induced photodegradation and the protective effect is significantly dependent on their concentration in the protein solution. There are no dramatic differences in the rates of exogenous and endogenous scavenging of generated free radicals for all concentrations used, with rate constants varying by a factor no greater than 2. The mechanism and the rate of scavenging or quenching are dependent on the nature of the radical species and the photostabilizer structure. Although this study provides evidence for free radical scavenging and protein protection, extrapolations to possible antioxidant effects in vivo must be made cautiously.
Similar content being viewed by others
REFERENCES
V. Nalini, B. Bax, H. Driessen, D. S. Moss, P. F. Lindley, and C. Slingsby (1994) J. Mol. Biol. 236, 1250.
H. Driessen, B. Bax, C. Slingsby, P. F. Lindley, D. Mahadevan, D. S. Moss, and I. Tickle (1991) Acta Crystallogr. Sect. B 47, 987.
G. Wistow (1993) Exp. Eye Res. 56, 729–732.
R. C. Augusteyn and J. Kortez (1987) FEBS Lett. 22, 1–5.
R. F. Borkman and J. McLaughlin (1995) Photochem. Photobiol. 62, 104–105.
J.-S. Lee, J.-H. Liao, S.-H. Wu, and S.-H. Chiou (1997) J. Protein Chem. 4, 283–289.
K. Mandal, S. K. Bose, and B. Chakrabarti (1986) Photochem. Photobiol. 43, 515–523.
Ch. Mohan Rao, D. Balasubramantan, and B. Chakrabarti (1987) Photochem. Photobiol. 46, 511–515.
R. J. W. Truscott and R. C. Augusteyn (1977) Biochim. Biophys. Acta 492, 43–52.
A. Balter (1991) Lens Eye Tox. Res. 8, 195–215.
K. Uchida, N. Enomoto, K. Itakura, and S. Kawakishi (1989) Agr. Biol. Chem. 53, 3285–3292.
J. N. Liang, S. K. Bose, and B. Chakrabarti (1985) Photochem. Photobiol. 40, 461–469.
U. P. Andley, P. Sutherland, J. N. Liang, and B. Chakrabarti (1984) Photochem. Photobiol. 40, 343–349.
S. K. Bose, K. Mandal, and B. Chakrabarti (1985) Biochem. Biophys. Res. Commun. 128, 1322–1328.
D. Y. Li, R. F. Borkman, R. H. Wang, and J. Dillon (1990) Exp. Eye Res. 51, 663–669.
J. D. Goosey, J. S. Zigler, and B. Chakrabarti (1985) Exp. Eye Res. 40, 461–469.
B. J. Ortwerth and P. R. Olsen (1994) Photochem. Photobiol. 60, 53–60.
B. J. Ortwerth, M. Linetsky, and P. R. Olesen (1995) Photochem. Photobiol. 62, 454–462.
H. O. Lowry, N. J. Rosebrough, A. L. Farr, and R. J. Randall (1951) J. Biol. Chem. 193, 265–275.
H. Bloemendal (1981) Molecular and Cellular Biology of the Eye, John Wiley and Sons, New York.
M. Linetsky and B. J. Ortwerth (1995) Photochem. Photobiol. 62, 87–93.
M. Linetsky and B. J. Ortwerth (1996) Photochem. Photobiol. 63, 649–655.
M. Linetsky and B. J. Ortwerth. (1997) Photochem. Photobiol. 65, 522–529.
S. Lerman (1980) Radiant Energy and the Eye, Macmillan, New York.
B. Halliwell and J. M. Gutteridge (1990) Methods Enzymol. 186, 1–85.
B. J. Ortwerth, A. Coots, H. J. James, and M. Linetsky (1998) Arch. Biochem. Biophys. 351, 189–196.
M. Linetsky, N. Ranson, and B. J. Ortwerth (1998) Arch. Biochem. Biophys. 351, 180–188.
T. P. Hum and R.C. Augusteyn (1987) Curr. Eye Res. 6, 1103.
J. A. Thomas, B. Poland, and R. Honzatko (1995) Biochim. Biophys. Acta 319, 1–9.
B. P. Lim, A. Nagao, J. Terao, K. Tanaka, T. Suzuki, and K. Takama. (1992) Biochim. Biophys. Acta 1126, 178–184.
Ch. S. Foote, Y. C. Chang, and R. W. Denny (1970) J. Am. Chem. Soc. 92, 5218–5219.
A. Taylor (1993) J. Am. Coll. Nutr. 12, 138–146.
S. T. Mayne (1996) FASEB J. 10, 690–701.
R. Ugarte, A. M. Edwards, M. S. Diez, A. Valenzuela, and E. Silva (1992) J. Photochem. Photobiol. B Biol. 13, 161–168.
J. Dillon (1991) J. Photochem. Photobiol. B Biol. 10, 23–40.
S. Lerman and R. Borkman (1977) Science 197, 1287–1288.
J. N. Liang, S. Bose, J. Thomson, and B. Chakrabarti (1988) Photochem. Photobiol. 47, 583–591.
K. Mandal, M. Kono, S. K. Bose, J. Thomson, and B. Chakrabarti (1988) Photochem. Photobiol. 4, 583–591.
J. N. Liang, S. K. Bose, and B. Chakrabarti (1985) Photochem. Photobiol. 40, 461–469.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kamińska, A., Kowalska, M. & Balter, A. A Comparative Study of the Effect of Exogenous and Endogenous Photostabilizers on Lens Crystallin Photodegradation. Journal of Fluorescence 9, 213–219 (1999). https://doi.org/10.1023/A:1022507701049
Issue Date:
DOI: https://doi.org/10.1023/A:1022507701049