Abstract
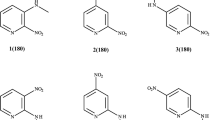
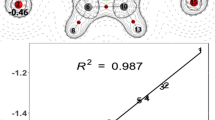
We have investigated the influence of volumetric (heterocyclic) substituents on the PMR characteristics of substituted nitroanilines depending on their position relative to the nitro group. Using quantum chemical simulation (the PM3 method), data on the position of the proton signals in the PMR spectra of 3-nitro-6-piperidinoaniline and 3-nitro-4-piperidinoaniline are interpreted. It is established that for structures in which the nitro group is removed from the benzene ring plane it does not appear possible to calculate the PMR spectrum using the Beeby–Sternhell increments.
Similar content being viewed by others
REFERENCES
Y. Igarashi, M. Shimoyamada, M. Takashima, and T. Suzuki, Method of Producing 2-amino-3-nitro-5-halogenopyrydine. U.S. Patent 5453512 (1995)
W. A. Denny, B. D. Palmer, and W. R. Wilson, Nitroaniline Derivatives and Their Use as Antitumor AgentsU.S. Patent 5571845 (1996).
Nitroaromatic Reduction. Chem. Market Report, No. 24, 5–13 (1993).
A. Lagrange, A. Junino, A. Genet, and I. Cofferet, Nouvelles 2-nitro-p-phenylenediamines hydroxyethylenes et leur utilisation en teinture des fibres keratiniques.France Patent 2692573 (1993).
R. Hamprecht and H. Jorden, Mischungen Blauer Dispersionsazofarbstoffe, Germany Patent Application 4437551 (1996).
H. Gunter, NMR Spectroscopy[Russian translation], Moscow (1984).
J. Beeby, S. Sternhell, T. Hoffmann-Ostenhof, E. Pretsch, and W. Simon, Anal. Chem., 45, No. 8, 1572–1582 (1973).
P. C. Begunov, V. Yu, Orlov, A. D. Kotov, V. V. Kopeikin, V. N. Leibzon, and G. S. Mironov, Izv. Vyssh. Uchebn. Zaved., Ser. Khim. Khim. Tekhnol., 41, Issue 4, 61–64 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Begunov, R.S., Orlova, T.N., Taranova, O.V. et al. Electronic Structure and PMR Characteristics of Substituted Nitroanilines. Journal of Applied Spectroscopy 69, 807–810 (2002). https://doi.org/10.1023/A:1022441932591
Issue Date:
DOI: https://doi.org/10.1023/A:1022441932591