Abstract
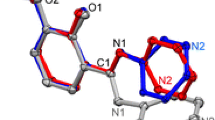
The para acetate ester azlactone of vanillin 2 was synthesized from vanillin 1 and hydrolyzed with sodium hydroxide. The yielded product 3 was investigated with X-ray Crystallographic and nuclear magnetic resonance techniques. Compound 3 crystallized in the orthorhombic Pbca space group (Z = 8) and with cell dimensions a = 14.732(2), b = 12.756(3), c = 12.747(6)Å revealing the enolate tautomer and not the keto form of 3-methoxy-4-hydroxyphenylpyruvic acid as the acetate ester. The structure exhibited the pyruvic acid side chain in the trans extended conformation. A single proton on the benzylic carbon atom further suggested the existence of the enolate tautomer form of 3 in solution. The chemical shift values and peak integration in the NMR spectra add additional support to this finding.
Similar content being viewed by others
References
Oliver, D.W.; Haasbroek, P.P.; Leger, J.M.; Carpy, A.J.M. J. Chem. Crystallogr. 1994, 24, 665.
Crooij, P. South African Patent Nr 68 03, 083, 1968.
MOLEN, An interactive Structure Solution Procedure, Enraf-Nonius, Delft, The Netherlands, 1990.
Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylor, R. J. Chem. Soc. Perkin Trans. II 1987, S1.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Haasbroek, P.P., Oliver, D.W. & Carpy, A.J.M. Enol tautomer of the acetate ester of 3-methoxy-4-hydroxyphenylpyruvic acid: Crystallographic and NMR spectroscopic evidence. Journal of Chemical Crystallography 28, 193–196 (1998). https://doi.org/10.1023/A:1022418210170
Issue Date:
DOI: https://doi.org/10.1023/A:1022418210170