Abstract
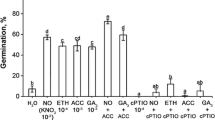
Methyl jasmonate (JA-Me) at 10−3 Minhibited Amaranthus caudatus seed germination anddecreased α-amylase activity. Exogenous gibberellin A3(GA3) and ethylene, but not benzyladenine (BA), increased activity ofthe enzyme in the presence of JA-Me, with ethylene being the most effective. Theinhibitor of ethylene action, 2,5-norbornadiene (NBD) inhibited seed germinationand decreased α-amylase activity. The inhibitory action of JA-Me onAmaranthus caudatus seed germination is associated with theinhibition of α-amylase activity. Exogenous GA3 and ethylenecontrol both α-amylase activity and seed germination in the presence of JA-Me.
Similar content being viewed by others
References
Akazawa T., Yamaguchi J. and Hayashi M. 1990. Rice α-amylase and gibberellin action-A personal view. In: Takahashi N., Phinney B.O. and MacMillan J. (eds), Gibberellins. Vol. 1. Springer-Verlag, New York, pp. 114–124.
Bia?ecka B. and Kępczyński J. 2002. The role of ethylene in reversing methyl jasmonate inhibition of Amaranthus caudatus seed germination induced by benzyladenine and gibberellins, (in preparation).
Black M., Corbineau F., Grzesik M., Guy P. and Come D. 1996. Carbohydrate metabolism in the developing and maturing wheat embryo in relation to its desiccation tolerance. J. Exp. Bot. 295: 161–169.
Chrispeels M.J. and Warner J.E. 1967. Gibberellic acid enhanced synthesis and release of α-amylase and ribonuclease by isolated barley aleurone layers. Plant Physiol. 42: 398–406.
Daletskaya T. and Sembdner G. 1989. Effect of jasmonic acid on germination of non-dormant and dormant seeds. Physiol. Rast. 36: 1118–1123.
Dunn G. 1974. A model for starch breakdown in higher plants. Phytochem. 13: 1341–1346.
El-Sayed S.T., Jwanny E.W., Rashad M.M., Mahmoud A.E. and Abdallah N.M. 1995. Glycosidases in plant tissues of some Brassicaceae. Applied Biochem. and Biotech. 55: 219–230.
Fincher G.B. 1989. Molecular and cellular biology associated with endosperm mobilization in germinating cereal grains. Annu. Rev. Plant Physiol. Plant Mol. Biol. 40: 305–346.
Hasegawa R., Maruyama A., Nakaya M., Tsuda S. and Esashi Y. 1995. The presence of two types of β-cyanoalanine synthase in germinating seeds and their responses to ethylene. Physiol. Plant. 93: 713–718.
Jacobsen J.V. and Chandler P.M. 1987. Gibberellin and abscisic acid in germinating cereals. In: Davies P.J. (ed.), Plant Hormones and Their Role in Plant Growth and Development. Martinus Nijhoff, Dordrecht, pp. 164–193.
Jones R.L. and Jacobsen J.V. 1991. Regulation of synthesis and transport of secreted proteins in cereal aleurone. Int. Rev. Cytol. 126: 49–88.
Kanzaki K., Kawabata C. and Noda K. 1993. Localization of α-amylase and its inhibitor in germinating wheat seed. Seed Sci. Research. 3: 287–292.
Kaur S., Gupta A.K. and Kaur N. 1998. Gibberellin A3 reverses the effect of salt stress in chickpea (Cicer arietinum L.) seedlings by enhancing amylase activity and mobilization of starch in cotyledons. Plant Growth Regul. 26: 85–90.
Kępczyński J. and Bia?ecka B. 1994. Stimulatory effect of ethephon, ACC, gibberellin A3 and A4 + 7 on germination of methyl jasmonate inhibited Amaranthus caudatus L. seeds. Plant Growth Regul. 14: 211–216.
Kępczyński J. and Bia?ecka B. 1997. The role of methyl jasmonate in germination of Amaranthus caudatus L. seeds. In: Ellis R.H., Black M., Murdoch A.J. and Hong T.D. (eds), Basic and Applied Aspects of Seed Biology. Kluwer Academic Publishers, Dordrecht, pp. 523–529.
Kępczyński J., Bia?ecka B. and Kepczynska E. 1999. Ethylene biosynthesis in Amaranthus caudatus seeds in response to methyl jasmonate. Plant Growth Regul. 28: 59–65.
Kępczyński J. and Karssen C.M. 1985. Requirement for the action of endogenous ethylene during germination of non-dormant seeds of Amaranthus caudatus. Physiol. Plant. 63: 49–52.
Lin C.C. 1984. Polyamine metabolism and relation to response of the aleurone layers of barley seeds to gibberellic acid. Plant Physiol. 74: 975–983.
Lin C.C. and Kao C.H. 1995. NaCl stress in rice seedlings: starch mobilization and the influence of GA3 on seedling growth. Bot. Bull. Acad. Sinica. 36: 169–173.
MacGregor A.W., Marchylo B.A. and Kruger J.E. 1988. Multiple α-amylase components in germinated cereal grains determined by isoelectric focusing and chromatofocusing. Cereal Chem. 65: 326–333.
Mitsui T. and Itoh K. 1997. The α-amylase multigene family. Trends Plant Sci. 2: 255–261.
Nolan R.C. and Ho T.H.D. 1988. Hormonal regulation of gene expression in barley aleurone layers. Planta. 174: 551–560.
Sugimoto N., Takeda G., Nagato Y. and Yamaguchi J. 1998. Temporal and spatial expression of the α-amylase gene during seed germination in rice and barley. Plant Cell Physiol. 39: 323–333.
Sun Z. and Henson C.A. 1991. A quantitative assessment of the importance of barley seed α-amylase, β-amylase, debranching enzyme, and α-glucosidase in starch degradation. Arch. Biochem. Biophys. 284: 298–305.
Tipirdamaz R., Durusoy M. and Bozcuk S. 1995. Effect of exogenous polyamines on α-amylase activity during seed germination under salt stress. Tr. J. of Bot. 19: 411–416.
Warner J.E., Ram Ch. and Chreespels M.J. 1965. Gibberellic acid controlled synthesis and release of-amylase in barley endosperm. J. Cell. Comp. Physiol. 66: 55–68.
Yamane H., Takagi H., Abe T., Yokata T. and Takahashi N. 1981. Identification of jasmonic acid in three species of higher plants and its biological activities. Plant Cell Physiol. 22: 689–697.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Białecka, B., Kępczyński, J. Regulation of α-amylase activity in Amaranthus caudatus seeds by methyl jasmonate, gibberellin A3, benzyladenine and ethylene. Plant Growth Regulation 39, 51–56 (2003). https://doi.org/10.1023/A:1021801504749
Issue Date:
DOI: https://doi.org/10.1023/A:1021801504749