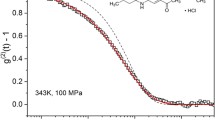
Abstract
Measurements of electrical conductivities of liquid and supercooled liquid NaCl–glycerol solutions were carried out between +42° and −87°C. Time-domain measurements of dielectric relaxation in pure glycerol and in NaCl–glycerol solutions between −78 and −89.9°C are described. The transient response data were fitted to the empirical Davidson–Cole response model. The specific conductivity data show a non-Arrhenius behavior near the glass-transition temperature and is well described by the Vogel–Tammann–Fulcher (VTF) law, which also fits the dielectric relaxation time data. The Vogel–Fulcher divergence temperature is consistent with the Kauzmann temperature. The dielectric relaxation time is increased significantly by the addition of sodium chloride, whereas the static relative permittivity decreases linearly with concentration, indicating that NaCl has a “structure-making” effect on the structure of glycerol.
Similar content being viewed by others
REFERENCES
H. Vogel, Phys. Z 22, 645 (1921).
G. Tammann and W. Hesse, Z. Anorg. Allg. Chem. 156, 245 (1926).
G. S. Fulcher, J. Am. Ceram. Soc. 8, 339 (1925).
M. R. Carpenter, D. B. Davies, and A. J. Matheson, J. Chem. Soc., Faraday Trans. II 69, 305 (1973).
D. W. Davidson and R. H. Cole, J. Chem. Phys. 19, 1484 (1951).
G. E. Mc Duffie and T. A. Litovitz, J. Chem. Phys. 37, 1699 (1962).
N. Menon and S. R. Nagel, Phys. Rev. Lett. 74, 1230 (1995).
P. K. Dixon, L. Wu, S. R. Nagel, B. D. Williams, and J. P. Carini, Phys. Rev. Lett. 65, 1108 (1990).
D. C. Champeney and F. O. Kaddour, Mol. Phys. 52, 509 (1984).
F. S. Howel, C. T. Moynihan, and P. B. Macedo, Bull. Chem. Soc. Jpn. 57, 652 (1984).
F. J. Bartoli, J. N. Birch, N. H. Toan, and G. E. McDuffie, J. Chem. Phys. 49, 1916 (1968).
N. O. Birge and S. R. Nagel, Phys. Rev. Lett. 54, 2674 (1985).
F. Accascina and S. Petrucci, Ricerca Scient. 29, 1640 (1959); S. Petrucci, Acta Chem. Scan. 16, 760 (1962).
E. H. Grant, R. J. Sheppard, and G. P. South, Dielectric Behavior of Biological Molecules in Solution, (Clarendon Press, Oxford, 1978).
N. R. Draper and H. Smith, Applied Regression Analysis, (Wiley, New York, 1981); E. S. Pearson and H. O. Hartley, Biometrica Tables for Statisticians Vol. 1, (Cambridge University Press, Cambridge, 1966).
P. K. Dixon, Phys. Rev. B 42, 8179 (1990).
R. V. Chamberlain, Phys. Rev. B 48, 15 638 (1993-I).
D. C. Champeney, R. N. Joarder, and J. C. Dore, Mol. Phys. 58, 337 (1986).
W. Kauzmann, Chem. Rev. 43, 219 (1948).
C. A. Angell and K. J. Rao, J. Chem. Phys. 57, 470 (1972).
C. A. Angell and D. L. Smith, J. Phys. Chem. 86, 3845 (1982).
F. A. Harris and C. T. O'konski, J. Phys. Chem. 61, 310 (1957).
A. Hammadi and D. C. Champeney, J. Solution Chem., in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hammadi, A., Champeney, D.C. Ionic Mobility and Dielectric Relaxation in Glass-Forming Mixtures of Sodium Chloride and Glycerol. Journal of Solution Chemistry 28, 21–34 (1999). https://doi.org/10.1023/A:1021747223332
Issue Date:
DOI: https://doi.org/10.1023/A:1021747223332