Abstract
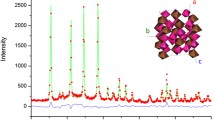
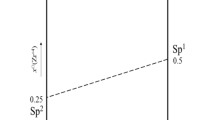
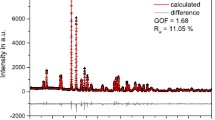
A powder X-ray diffraction experiment was performed on cubic Zr1-xHfxW2O8 (x=0.25, 0.50 and 0.75) solid solutions from 90 to 560 K. The lattice parameters of Zr1-xHfxW2O8 at 121 K decreased linearly with increasing Hf contents, due to smaller ionic radius of hafnium than that of zirconium. Transition temperatures due to α-β structural phase transition increased with increasing Hf contents, reflecting the decrease of lattice free volume related to the orientation of unshared vertex of WO4. Anomaly in the heat capacity of Zr0.5Hf0.5W2O8 was observed around 450 K which was 9 K lower than that by X-ray diffraction method. Transition entropy of Zr0.5Hf0.5W2O8 was 2.1 J mol-1 K-1, consistent with those of ZrW2O8 and HfW2O8. This consistent entropy supports that Zr1-xHfxW2O8 (x=0-1.0) has the same order-disorder phase transition mechanism.
Similar content being viewed by others
References
T. A. Mary, J. S. O. Evans, T. Vogt and A. W. Sleight, Science, 272 (1996) 90.
J. S. O. Evans, T. A. Mary, T. Vogt, M. A. Subramanian and A. W. Sleight, Chem. Mater., 8 (1996) 2809.
J. S. O. Evans, W. I. F. David and A. W. Sleight, Acta Crystallogr., Sect. B: Struct. Sci., 55 (1999) 333.
A. P. Ramirez and G. R. Kowach, Phys. Rev. Lett., 80 (1998) 4903.
G. Ernst, C. Broholm, G. R. Kowach and A. P. Ramirez, Nature, 396 (1998) 147.
A. W. Sleight, T. A. Mary and J. S. O. Evans, U.S. Patent number 5514360, 1995.
Y. Yamamura, N. Nakajima and T. Tsuji, Solid State Commun., 114 (2000) 453.
Y. Yamamura, N. Nakajima and T. Tsuji, Phys. Rev. B, 64 (2001) 184109.
D. G. Archer, J. Phys. Chem. Ref. Data, 22 (1993) 1441.
J. B. Nelson and D. P. Riley, Proc. Phys. Soc., 57 (1945) 160.
R. D. Shannon, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., A32 (1976) 751.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakajima, N., Yamamura, Y. & Tsuji, T. Phase transition of negative thermal expansion Zr1-xHfxW2O8 solid solutions. Journal of Thermal Analysis and Calorimetry 70, 337–344 (2002). https://doi.org/10.1023/A:1021647717948
Issue Date:
DOI: https://doi.org/10.1023/A:1021647717948