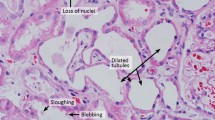
Abstract
Indinavir sulfate is a protease inhibitor used of the treatment of primary HIV infection either as monotherapy or as part of antiretroviral treatment schemes. Approximately 10% of all patients develop urolithiasis with radiolucent stones consisting of indinavir.We present our results of the treatment in 11 HIV positive patients (9 men, 2 women), who developed Indinavir lithiasis after 5–8 months of antiretroviral therapy. Following the initial procedures (spasmoanalgetic drugs, ureteroscopy, double J-stent or nephrostomy), the patients were further treated by increasing diuresis and urinary acidification.All the patients responded well to the treatment, the obstruction was releieved and their renal function was restored to normal.
Similar content being viewed by others
References
Brzosko S, Hryszko T, Panasiuk B et al. Nephrolithiasis in an HIV infected patient treated with indinavir. Przegl Lek 2001; 58(6): 538–539.
Chen IW, Vastag KJ, Lin JH. High-performance liquid chromatographic determination of a potent and selective HIV protease inhibitor (L-735, 524) in rat dog and monkey plasma. J Chromatography 1995; B 672: 111–117.
Crixivan® package insert. Merck and Co., Inc., US Human Health 1996.
Daudon M, Estépa L, Viard JP et al. Urinary stones in HIV-1-postitive patients treated with indinavir. The Lancet 1997 May; 349: 1294–1295.
Gendle DL, Stoller ML, Jarrett TW et al. Protease inhibitor-induced urolithiasis. Urology 1997; 50: 508–511.
Godwin TA. HIV/AIDS case histories: Indinavir crystalluria. AIDS Patient Care STDS 2001 Mar; 15(3): 169–171.
Grady BR, Munch LC, Hoven AD et al. Urolithiasis associated with the protease inhibitor indinavir. Urology 1997; 50(4): 513–518.
Gulick R, Mellors J, Havlur D. Potent and sustained antiretroviral activity of indinavir (IDV) in combination with zidovudine (ZVD) and lamivudine (3TC). 3rd Conference on Retroviruses and Opportunistic Infection 1996; Washington (Abstract): 162.
Massari F, Stazewski S, Berry P. A double-blind randomised trial of indinavir (MK-639) alone or with zidivudine vs. zidivudine alone in zidivudinenaï ve patients. 35th Interscience Conference on Antimicrobial Agents and Chemotherapy 1995; California (Abstract): LB-6.
New drugs for HIV infection. Med Let Drugs Ther 1996; 38: 35–37.
Reilly RF, Tray K, Perazella MA. Indinavir nephropathy revisited: a pattern of insidious renal failure with identifiable risk factors. Am J Kidney Dis 2001 Oct; 38(4): E23.
Salahuddin S, Hsu YS, Bucholz NP et al. Is indinavir crystalluria an indicator for indinavir stone formation. AIDS 2001 May 25; 15(8): 1079–1080.
Tashima KT, Horowitz JD, Rosen S. Indinavir nephropathy. N Engl J Med 1997; 336: 138–140.
Vacca JP, Dorsey PD, Schleif WA et al. L-735, 524: an orally bioavailable human immunodeficiency virus type 1 protease inhibitor. Proc Natl Acad Sci USA 1994; 91: 4096–4100.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalaitzis, C., Dimitriadis, G., Touloupidis, S. et al. Treatment of Indinavir sulfate induced urolithiasis in HIV-positive patients. Int Urol Nephrol 34, 13–15 (2002). https://doi.org/10.1023/A:1021340915465
Issue Date:
DOI: https://doi.org/10.1023/A:1021340915465