Abstract
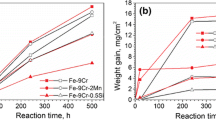
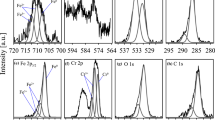
The corrosion reaction of four Fe–Mn–Al alloys exposed to a cycling, dry–humid, SO2 (0.001% by volume) polluted atmosphere was studied. ICEMS, XPS, AES-SAM and transmission Mössbauer spectroscopy at different temperatures were employed to characterize the corrosion products. The analytical results indicate that (i) ferrihydrite is the main component of the rust; (ii) there is an abundant presence of Mn2+ and SO3 2−/SO4 2− on the top of the corrosion layer, the concentration of SO4 2− increasing with the number of cycles; and (iii) the magnetic hyperfine pattern exhibited by the series of low-temperature spectra of the rust is quite different from that observed in the rust formed under similar corrosive environments on iron and weathering steel. This latter finding is correlated with a slow rate of transformation of the Fe3+ species formed at the early stages of corrosion into α-FeOOH, the usual final product of this type of corrosion processes. The sulphate anions, abundant inside the electrolyte during the wet periods, could be incorporated to the ferrihydrite structure being responsible for the Mössbauer spectral pattern recorded from the corrosion products at low temperatures.
Similar content being viewed by others
References
Altstetter, C. J., Bentley, A. P., Fourie, J.W. and Kirkbride, A. N., Mat. Sci. Eng. 82 (1986), 13.
Agudelo, A. C., Marco, J. F., Gancedo, J. R., Gracia, M. and Pérez-Alcázar, G. A., Hyp. Interact. (C) (1998).
Marco, J. F., Dávalos, J. Z., Gracia, M. and Gancedo, J. R., Hyp. Interact. 83 (1994), 111.
Marco, J. F., Dávalos, J. Z., Gracia, M. and Gancedo, J. R., Hyp. Interact. 46 (1989), 453.
Murad, E. and Johnston, J. H., In: G. J. Long (ed.), Mössbauer Spectroscopy Applied to Inorganic Chemistry, Vol. 2, Plenum Press, New York, 1987, p. 507.
Murad, E., Bigham, J. M., Bowen, L. H. and Shvertmann, U., Hyp. Interact. 58 (1990), 2373.
Nalovic, L. and Janot, C., Rev. de Physique Appliqué 14 (1979), 475.
Bowen, L. H., De Grave, E. and Vandenberghe, R. E., In: G. J. Long and F. Grandjean, (eds), Mössbauer Spectroscopy Applied to Magnetism and Materials Science, Vol. 1, Plenum Press, New York, 1993, p. 115.
Pourbaix, M., Atlas d'equilibres electrochemiques, Gauthier-Villarrs, Paris, 1963, p. 171.
Leidheiser Jr., H. and Music, S., Corros. Sci. 22 (1982), 1089.
Oku, M., Hirokawa, K. and Ikeda, S.,J. Electron Spectrosc. Relat. Phenom. 7 (1975), 465; Brisk, M. A. and Baker, A. D., J. Electron Spectrosc. Rel. Phenom. 7 (1975), 197.
Allen, C., Harris, S. J., Jutson, J. A. and Dyke, J. M., Appl. Surf. Sci. 37 (1989), 111.
McIntyre, N. S. and Zetaruk, D. G., Anal. Chem. 49 (1977), 1521.
Choudhury, T., Saied, S. O., Sullivan, J. L. and Abbot, A. M., J. Phys. D 22 (1989), 1185.
Agudelo, A. C., Gancedo, J. R., Marco, J. F. and Hanzel, D., J. Vac. Sci. Technol. A 15 (1997), 3163.
Berger, F., Beche, E., Berjoan, R., Klein, D. and Chambaudet, A., Appl. Surf. Sci. 93 (1996), 9.
Van der Giessen, A. A., J. Inorg. Nucl. Chem. 28 (1966), 2155.
van der Kraan, A. M. and Medema, J., J. Inorg. Nucl. Chem. 31 (1969), 2039.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Agudelo, A.C., Marco, J.F., Gancedo, J.R. et al. Fe–Mn–Al–C Alloys: a Study of Their Corrosion Behaviour in SO2 Environments. Hyperfine Interactions 139, 141–152 (2002). https://doi.org/10.1023/A:1021248519209
Issue Date:
DOI: https://doi.org/10.1023/A:1021248519209