Abstract
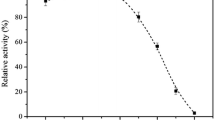
When grown on a sucrose-containing medium, Candida utilis synthesizes and secretes two invertases: one of molecular size of 280 kDa (the S-form – Slow-migrating) and a new form of Mr of 62 kDa (the F-form – Fast-migrating). Prior to immobilization, purification of S- and F-forms of invertase increased the immobilization yield to 89–100%, in comparison with that of crude invertase preparation (52%). The immobilized purified S- and F-form of invertase remained partially active after 15 min at 100 °C; the F-form retained almost 30% of its maximum activity. The immobilized S-form or F-form of invertase almost completely inverted (95% hydrolysis) 60% (w/v) sucrose over 5 h continuous reaction at 80 °C. Moreover, at 90 °C the immobilized F-form hydrolysed 70% of 60% (w/v) sucrose over 5 h, while the capability of the immobilized S-form of inverting sucrose over 5 h reaction decreased from 80% to 45%.
Similar content being viewed by others
References
Aksu Z, Kutsal T (1986) Lactic acid production from molasses utilizing Lactobacillus delbrueckii and invertase together. Biotechnol. Lett. 8: 157–160.
Belcarz A, Ginalska G, Lobarzewski J, Penel C (2002) The novel, non-glycosylated invertase form Candida utilis (conditions of biosynthesis, purification and properties). Biochim. Biophys. Acta 1594: 40–53.
Boze H, Moulin G, Galzy P (1994) Production of food and fodder yeast. Crit. Rev. Biotechnol. 12: 65–86.
Chavez FP, Rodriguez L, Diaz J, Delgado JM, Cremata JA (1997) Purification and characterization of an invertase from Candida utilis: comparison with natural and recombinant yeast invertases. J. Biotechnol. 53: 67–74.
Chiang CJ, Lee WC (1997) Immobilization of ?-fructofuranosidases from Aspergillus on methacrylamide-based polymeric beads for production of fructooligosaccharides. Biotechnol. Progr. 13: 577–582.
Ginalska G, Belcarz A, Lobarzewski J, Wolski T (1999) Application of immobilized invertase for continuous production of inverted sucrose. Biotechnologia 1: 276–237.
Huang CT, Lee WC (1994) Immobilization of invertase onto a copolymer of methacrylamide and N,N_-methlenebis(acrylamide). Appl. Biochem. Biotechnol. 45/46: 663–672.
Husain S, Jafri F, Saleemuddin M (1996) Effects of chemical modification on the stability of invertase before and after immobilization. Enzyme Microb. Technol. 18: 275–280.
Iizuka M, Yamamoto T (1972) Studies on invertase of Candida utilis. Part I. Comparative study on isolation methods of the enzyme. Agr. Biol. Chem. Tokyo 36: 349–355.
Iizuka M, Tsuji Y, Yamamoto T (1974) Some physicochemical properties of invertase of Candida utilis. Agr. Biol. Chem. Tokyo 38: 213–215.
Kurokawa Y, Suzuki K, Tamai Y (1998) Adsorption and enzyme (β-galactosidase and ?-chymotrypsin): immobilization properties of gel fiber prepared by the gel formation of cellulose acetate and titanium iso-propoxide. Biotechnol. Bioeng. 59: 651–656.
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Lloyd JB, Whelan WJ (1969) An improved method for enzymic determination of glucose in the presence of maltose. Anal. Biochem. 30: 467–490.
Mansfield J, Forster M, Hoffmann T, Schellenberger A (1995) Coimmobilization of Yarrowia lipolytica cells and invertase in polyelectrolyte complex microcapsules. Enzyme Microb. Technol. 17: 11–17.
Monsan P, Combes D (1984) Application of immobilized invertase to continuous hydrolysis of concentrated sucrose solutions. Biotechnol. Bioeng. 26: 347–351.
Naundorf A, Caussette M, Ajisaka K (1998) Characterization of the immobilized β-galactosidase C from Bacillus circulans and the production of β-(1-3)-linked disaccharides. Biosci. Biotech. Bioch. 62: 1313–1317.
Nikiel S (1996) Sugar Industry, 3rd edn. Warszawa, Poland: WSP.
Schacterle GR, Pollack RL (1973) A simplified method for the quantitative assay of small amounts of protein in biologic material. Anal. Biochem. 51: 654–655.
Selampinar F, Akbulut U, Ozden MY, Toppare L (1997) Immobilization of invertase in conducting polymer matrices. Biomaterials 18: 1163–1168.
Tull D, Gottschalk TE, Svendsen I, Kramhøft B, Phillipson BA, Bisgärd-Frantzen H, Olsen O, Svensson B (2001) Extensive N-glycosylation reduces the thermal stability of a recombinant alkalophilic Bacillus ?-amylase produced in Pichia pastoris. Protein Expres. Purif. 21: 13–23.
Weethall H (1976) Covalent coupling methods for inorganic support material. Method Enzymol. 44: 134–148.
Zaborsky OR, Ogletree J (1974) The immobilization of glucose oxidase via activation of its carbohydrate residues. Biochem. Biophys. Res. Co. 61: 210–216.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Belcarz, A., Ginalska, G. & Lobarzewski, J. Immobilized, thermostable S- and F-forms of the extracellular invertase from Candida utilis can hydrolyse sucrose up to 100 °C. Biotechnology Letters 24, 1993–1998 (2002). https://doi.org/10.1023/A:1021138101897
Issue Date:
DOI: https://doi.org/10.1023/A:1021138101897