Abstract
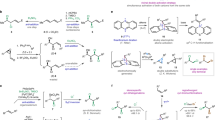
When treated with KOH under phase-transfer catalysis or with ButOK, 3-substituted (alkyl or phenyl) 1,1,3-tribromo-1-fluoropropanes 1a—c exclusively generate previously unknown (alk-1-ynyl)fluorocarbenes 5a—c, which react with olefins to give 1-(alk-1-ynyl)-1-fluorocyclopropanes 6a—h in 12—69% yields. Under analogous conditions, 3-alkyl- and 3-aryl-3-bromo-1,1,1-trichloropropanes 2a—c selectively afford (alk-1-ynyl)chlorocarbenes 7a—c, which are trapped by olefins to form the corresponding 1-(alk-1-ynyl)-1-chlorocyclopropanes 8a—k in 35—70% yields. (Phenylethynyl)chlorocarbene 7a is also selectively generated from 1,1,1,3-tetrachloro-3-phenylpropane (3a) upon its treatment with ButOK. With an excess of 2,3-dimethylbut-2-ene or 2-methylpropene, carbene 7a yields 1-chloro-1-(phenylethynyl)cyclopropanes 8a or 8c, respectively. In contrast, 1,1,1,3-tetrachloroheptane 3b and 3-alkyl- and 3-phenyl-1,1,1,3-tetrabromopropanes 4a,c,f react with bases in the presence of olefins to give, along with the corresponding 1-(alk-1-ynyl)-1-halocyclopropanes 8a,c,d and 11a—f, vinylidenecyclopropanes 12a,c—g, which suggests the generation, under these conditions, both (alk-1-ynyl)halocarbenes 7b and 9a—c and vinylidenecarbenes 10 and 11a—c. The composition and structures of intermediate products in the reactions of tetrahalides 1b, 2a, 2b, 3a, and 3b with ButOK were determined and the mechanisms for carbene generation in these reactions were proposed.
Similar content being viewed by others
References
K. N. Shavrin, V. D. Gvozdev, and O. M. Nefedov, Izv. Akad. Nauk, Ser. Khim., 1998, 1185 [Russ. Chem. Bull., 1998, 47, 1154 (Engl. Transl.)].
K. N. Shavrin, V. D. Gvozdev, and O. M. Nefedov, Mendeleev Commun., 1997, 144.
K. N. Shavrin, V. D. Gvozdev, and O. M. Nefedov, Izv. Akad. Nauk, Ser. Khim., 1997, 2079 [Russ. Chem. Bull., 1997, 46, 1973 (Engl. Transl.)].
K. N. Shavrin, V. D. Gvozdev, and O. M. Nefedov, Izv. Akad. Nauk, Ser. Khim., 1992, 1128 [Bull. Russ. Acad. Sci., Div. Chem. Sci., 1992, 41, 885 (Engl. Transl.)].
K. N. Shavrin, I. V. Krylova, I. B. Shvedova, G. P. Okonnishnikova, I. E. Dolgy, and O. M. Nefedov, J. Chem. Soc., Perkin Trans. 2, 1991, 1875.
K. N. Shavrin, I. B. Shvedova, and O. M. Nefedov, Izv. Akad. Nauk SSSR, Ser. Khim., 1991, 2559 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1991, 40, 2235 (Engl. Transl.)].
M. S. Kharash, O. Reinmuth, and W. H. Urry, J. Am. Chem. Soc., 1947, 69, 1105.
M. Asscher and D. Vosfi, J. Chem. Soc., 1963, 1887.
H. Günther, NMR Spectroscopy, an Introduction, J. Wiley and Sons, Chichester-New York-Brisbane-Toronto, 1972.
Matsui Kiyohide, Negishi Akira, Takahatake Yuriko, Sugimoto Kikuo, Fujimoto Tamotsu, Takashima Toshiyuki, and Kondo Kiyosi, Bull. Chem. Soc. Jpn., 1986, 59, 221.
R. A. Moss, Acc. Chem. Res., 1980, 13, 58.
H. D. Harzler, J. Am. Chem. Soc., 1961, 83, 4990.
P. J. Stang, in Methoden der organischen Chemie (Houben-Weyl), Stuttgart, Georg Thieme, 1989, B. E19b, Teil 1, 138.
A. Roedig, in Methoden der organischen Chemie (Houben-Weyl), Stuttgart, Georg Thieme, 1960, B. V/4, 773.
J. M. Birchall and R. N. Hasseldine, J. Chem. Soc., 1956, 16.
G. Lehnmann, J. Prakt. Chem., 1963, 22, 230.
A. J. Gordon and R. A. Ford, The Chemist´s Companion, J. Wiley and Sons, New York-London-Sydney-Toronto, 1972.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shavrin, K.N., Gvozdev, V.D. & Nefedov, O.M. Alkynylhalocarbenes. 4. Generation of (alk-1-ynyl)halocarbenes from 3-substituted 1,1,1,3-tetrahalopropanes under the action of bases. Russian Chemical Bulletin 51, 1237–1253 (2002). https://doi.org/10.1023/A:1020952513679
Issue Date:
DOI: https://doi.org/10.1023/A:1020952513679