Abstract
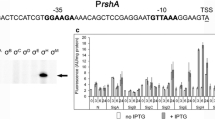
The presumed DNA target for the Rhodobacter sphaeroides 2.4.1 FnrL regulatory protein is the FNR consensus sequence, TTGAT-N4-ATCAA, based on (1) similarities between the helix-turn-helix motifs of FnrL and the Escherichia coli homologue, Fnr, and (2) the established FnrL dependence for anaerobic induction of six gene clusters all having upstream FNR consensus-like sequences. We are interested in understanding the regulation of one among these; namely, the hemA gene, which codes for one of two isozymes of 5-aminolevulinate (ALA) synthase in this organism. Here, we present in vivo evidence that the hemA gene is transcribed from two promoters. Both are under oxygen control, and disabling the fnrL gene abolishes induction of each promoter in response to lowering oxygen tension. Based on the 5′ position of the FNR consensus sequence relative to the downstream promoter, we had hypothesized that activation of that promoter is mediated by binding of FnrL to the FNR consensus sequence. Consistent with this hypothesis, we found here that transcription from the downstream promoter is no longer inducible when the FNR consensus sequence is deleted. With respect to the upstream promoter, based on the fact that the +1 site of transcription from that promoter is within the FNR consensus sequence, we propose an indirect role for FnrL. This possibility is discussed, together with other unresolved aspects of hemA expression.
Similar content being viewed by others
References
Altschul S, Madden T, Schäffer A, Zhang J, Zhang Z, Miller W and Lipman D (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25: 3389–3402
Boyer H and Roulland-Dussoix D (1969) A complementation analysis of the restriction and modification of DNA in Escherichia coli. J Mol Biol 41: 459–472
Davis J, Donohue TJ and Kaplan S (1988) Construction, characterization, and complementation of a Puf? mutant of Rhodobacter sphaeroides. J Bacteriol 170: 320–329
Ditta G, Stanfield S, Corbin D and Helinski D (1980) Broad host range DNA cloning system for gram negative bacteria: construction of a gene bank of Rhizobium meliloti. Proc Natl Acad Sci USA 77: 7347–7351
Donohue TJ, McEwan AG and Kaplan S (1986) Cloning, DNA sequence, and expression of the Rhodobacter sphaeroides cytochrome c 2 gene. J Bacteriol 168: 962–972
Eraso JM and Kaplan S (1994) prrA, a putative response regulator involved in oxygen regulation in photosynthesis gene expression in Rhodobacter sphaeroides. J Bacteriol 176: 32–43
Kiley P and Kaplan S (1987) Cloning, DNA sequence, and expression of the Rhodobacter sphaeroides light-harvesting B800-850-? and B800-850-? genes. J Bacteriol 169: 3268–3275
Lee J and Kaplan S (1995) Transcriptional regulation of puc operon expression in Rhodobacter sphaeroides. Analysis of the cis-acting downstream regulatory sequence. J Biol Chem 270: 20453–20458
Meinhardt SW, Kiley PJ, Kaplan S, Crofts AR and Harayama S (1985) Characterization of light-harvesting mutants of Rhodopseudomonas sphaeroides. I. Measurement of the efficiency of light energy transfer from light-harvesting complexes to the reaction center. Arch Biochem Biophys 236: 130–139
Mouncey NJ and Kaplan S (1998a) Oxygen regulation of the ccoN gene encoding a component of the cbb3 oxidase in Rhodobacter sphaeroides 2.4.1T: involvement of the FnrL protein. J Bacteriol 180: 2228–2231
Mouncey NJ and Kaplan S (1998b) Cascade regulation of dimethyl sulfoxide reductase (dor) gene expression in the facultative phototroph Rhodobacter sphaeroides 2.4.1T. J Bacteriol 180: 2924–2930
Neidle EL and Kaplan S (1993) Expression of the Rhodobacter sphaeroides hemA and hemT genes, encoding two 5-aminolevulinic acid synthase isozymes. J Bacteriol 175: 2292–2303
Oh J-I, Eraso J and Kaplan S (2000) Interacting regulatory circuits involved in orderly control of photosynthesis gene expression in Rhodobacter sphaeroides 2.4.1. J Bacteriol 182: 3081–3087
Sambrook J, Fritsch E and Maniatis T (1985) Molecular Cloning: a Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Sistrom WR (1960) A requirement for sodium in the growth of Rhodopseudomonas sphaeroides. J Gen Microbiol 22: 778–785
Tai T-N, Havelka WA and Kaplan S (1988a) A broad-host-range vector system for cloning and translational lacZ fusion analysis. Plasmid 19: 175–188
Tai T-N, Moore MD and Kaplan S (1988b) Cloning and characterization of the 5-aminolevulinate synthase gene(s) from Rhodobacter sphaeroides. Gene 70: 139–151
van Neil CB (1944) The culture, general physiology, and classification of the non-sulfur purple and brown bacteria. Bacteriol Rev 8: 1–118
Zeilstra-Ryalls JH, Gabbert K, Mouncey NJ, Kranz RG and Kaplan S (1997) Analysis of the fnrL gene and its function in Rhodobacter capsulatus. J Bacteriol 179: 7264–7273
Zeilstra-Ryalls JH and Kaplan S (1995a) Regulation of 5-aminolevulinic acid synthesis in Rhodobacter sphaeroides 2.4.1: the genetic basis of mutant H-5 auxotrophy. J Bacteriol 177: 2760–2768
Zeilstra-Ryalls JH and Kaplan S (1995b) Aerobic and anaerobic regulation in Rhodobacter sphaeroides 2.4.1: the role of the fnrL gene. J Bacteriol 177: 6422–6431
Zeilstra-Ryalls JH and Kaplan S (1998) Role of the fnrL gene in photosystem gene expression and photosynthetic growth of Rhodobacter sphaeroides 2.4.1. J Bacteriol 180: 1496–1503
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fales, L., Nogaj, L. & Zeilstra-Ryalls, J. Analysis of the upstream sequences of the Rhodobacter sphaeroides 2.4.1 hemA gene: in vivo evidence for the presence of two promoters that are both regulated by fnrL * . Photosynthesis Research 74, 143–151 (2002). https://doi.org/10.1023/A:1020947308227
Issue Date:
DOI: https://doi.org/10.1023/A:1020947308227