Abstract
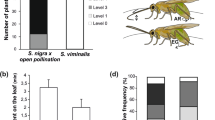
Yponomeuta cagnagellus is a phytophagous moth species specialized on Euonymus europaeus. Host discrimination by the adult female is an important aspect of host specialization and is based mainly on the distinctive secondary chemistry of host and nonhosts. This paper describes a bioassay that was developed to study the effect of isolated plant surface compounds on Yponomeuta oviposition. Adult moths recognize their hosts through chemical stimuli on the leaf or twig surface. Relatively apolar compounds extracted from the host twig surface by washing in dichlormethane do not stimulate oviposition. More polar, methanol-soluble compounds do, and this stimulation is dose dependent. Moths are able to recognize hosts solely by their surface compounds: females show a strong preference for artificial twigs treated with methanolic extracts of their hosts compared to those treated with methanolic extracts of nonhosts Crataegus monogyna and Prunus spinosa (both of which are hosts for closely related Y. padellus). Shape and surface characteristics of the oviposition substrate also influence oviposition. The substrate needs to resemble the basic form of a twig (i.e., cylindrical), and females prefer a coarse surface with irregularities over a smooth one.
Similar content being viewed by others
REFERENCES
Berenbaum, M. R. 1990. Evolution of specialization in insect-umbellifer associations. Annu. Rev. Entomol. 35:319-343.
Bordner, J., Danehower, D. A., Thacker, J. D., Kennedy, G. G., Stinner, R. E., and Wilson, K. G. 1983. Chemical basis of host plant selection, pp. 245-264, in P. A. Hedin (ed.). Plant Resistance to Insects. ACS Symposium Series. American Chemical Society, Washington, D.C.
Bremner, E., Hora, K. H., and Roessingh, P. 1997. Oviposition in Yponomeuta cagnagellus: When, where and why there? Proc. Exp. Appl. Entomol. 8:183-188.
Burton, R. L., and Schuster, D. J. 1981. Oviposition stimulant for tomato pinworms from surfaces of tomato plants. Ann. Entomol. Soc. Am. 74:512-515.
Bush, G. L. 1994. Sympatric speciation in animals: New wine in old bottles. Trends Ecol. Evol. 9:285-288.
Derridj, S., Fiala, V., Barry, P., Robert, P., Roessingh, P., and StÄdler, E. 1992. Role of nutrients found in the phylloplane, in the insect host-plant selection for oviposition, pp. 139-140, in S. B. J. Menken, J. H. Visser, and P. Harrewijn (eds.). Proceedings, 8th Symposium on Insect-Plant Relationships. Kluwer Academic, Dordrecht.
Dethier, V. G. 1982. Mechanisms of host-plant recognition. Entomol. Exp. Appl. 31:49-56.
Gershenson, Z. S., and S. A. Ulenberg. 1998. The Yponomeutinae (Lepidoptera) of the world exclusive of the Americas. Verh. K. Ned. Akad. Wet. Afd. Natuurk. Tweede Reeks. 99:1-202.
Hendrikse, A. 1979. Activity patterns and sex pheromone specificity as isolating mechanisms in eight species of Yponomeuta (Lepidoptera: Yponomeutidae). Entomol. Exp. and Appl. 25:172-178.
Honda, K. 1990. Identification of host-plant chemicals stimulating oviposition by swallowtail butterfly, Papilio protenor. J. Chem. Ecol. 16:325-337.
Honda, K. 1995. Chemical basis of differential oviposition by lepidopterous insects. Arch Insect Biochem. Physiol. 30:1-23.
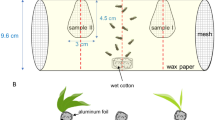
Hora, K. H., and Roessingh, P. 1999. Oviposition in Yponomeuta cagnagellus: The importance of contact cues for host plant acceptance. Physiol. Entomol. 24:109-120.
Janz, N., and Nylin, S. 1997. The role of female search behavior in determining host plant range in plant feeding insects: A test of the information processing hypothesis. Proc. R. Soc. London B 264:701-707.
Menken, S. B. J., and Roessingh, P. 1998. Evolution of insect-plant associations: Sensory perception and receptor modifications direct food specialization and host shifts in phytophagous insects, pp. 145-156, in D. Howard and S. H. Berlocher (eds.). Endless Forms: Species and Speciation, Academic Press, New York.
Menken, S. B. J., Herrebout, W. M., and Wiebes, J. T. 1992. Small ermine moths (Yponomeuta): Their Host Relations and Evolution. Annu. Rev. Entomol. 37:41-66.
Ramaswamy, S. B. 1988. Host finding by moths: Sensory modalities and behaviours. J. Insect Physiol. 34:235-249.
Renwick, J. A. A., and Chew, F. S. 1994. Oviposition behavior in Lepidoptera. Annu. Rev. Entomol. 39:377-400.
Singer, M. C. 1984. Butterfly-hostplant relationships: Host quality, adult choice and larval success, pp. 81-88, in R. I. Vane-Wright and P. Ackery (eds.). The Biology of Butterflies, Vol. 13, Academic, London.
Soldaat, I. I., Boutin, J., and Derridj, S. 1996. Species-specific composition of free amino acids on the leaf surface of four Senecio species. J. Chem. Ecol. 22:1-12.
StÄdler, E., and Roessingh, P. 1991. Perception of surface chemicals by feeding and ovipositing insects. Symp. Biol. Hung. 39:71-86.
Thibout, E., and Auger, J. 1996. Behavioural events and host constituents involved in oviposition in the leek moth Acrolepiopsis assectella. Entomol. Exp. Appl. 80:101-104.
Udayagiri, S., and Mason, C. E. 1995. Host plant constituents as oviposition stimulants for a generalist herbivore: European corn borer. Entomol. Exp. Appl. 76:59-65.
Wiklund, C. 1975. The evolutionary relationship between adult oviposition preferences and larval host plant range in Papilio machaon L. Oecologia 18:185-197.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hora, K.H., Roessingh, P. A Novel Bioassay for Yponomeuta cagnagellus Oviposition in Response to Extracts of Host and Nonhost Plant Surface Compounds. J Chem Ecol 25, 2547–2559 (1999). https://doi.org/10.1023/A:1020882326462
Issue Date:
DOI: https://doi.org/10.1023/A:1020882326462