Abstract
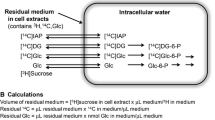
Hexokinase (HK, EC 2.7.1.1) is a key enzyme in the control of brain glucose metabolism. The regulatory role of HK in different neural cell types has not been elucidated. In this study we determined some kinetic and regulatory properties of HK in mouse cerebrocortical astrocytes in primary culture. Astroglial HK showed an absolute requirement for Mg-ATP and D-glucose. The pH optimum of HK was between 7.4 and 8.0. For astroglial HK, the Km for Mg-ATP was ~ 208 μM and Vmax ~ 35.4 mU/mg protein. At levels higher than 0.2 mM, D-glucose-1,6-bisphosphate, a known regulator of glycolysis, inhibited astroglial HK in a concentration-dependent manner, with an IC50 of ~ 0.4 mM; at 1.2 mM, it almost completely inhibited HK activity. The results obtained for astroglial HK are compatible with those reported for the highly purified preparations of brain HK. These data are of direct relevance to the assessment of glycolytic flux and its regulation in astrocytes.
Similar content being viewed by others
REFERENCES
Beitner, R. (1990). Regulation of carbohydrate metabolism by glucose 1,6-bisphosphate in extrahepatic tissues; comparison with fructose 2,6-bisphosphate. Int. J. Biochem. 22:553–557.
Beitner, R., Klein, S., and Nordenberg, J. (1982). The participation of glucose-1,6-diphosphate in the regulation of hexokinase and phosphoglucomutase activities in brains of young and adult rats. Int. J. Biochem. 14:195–199.
Butterworth, R.F. (1989). Enzymes of the pyruvate dehydrogenase complex of mammalian brain. In (A.A. Boulton, G.B. Baker, and R.F. Butterworth (eds.), Neuromethods, Vol. 11, Humana Press, Clifton, NJ, pp. 283–307.
Clark, J.B., and Lai, J.C.K. (1989). Glycolytic, tricarboxylic acid cycle, and related enzymes in brain. In (A.A. Boulton, G.B. Baker, and R.F. Butterworth (eds.), Neuromethods, Vol. 11, Humana Press, Clifton, NJ, pp. 233–281.
Dombrowski Jr., G.J., Swiatek, K.R., and Chao, K.-L. (1994). Hexose diphosphate and phosphofructokinase in rat brain during development. Neurochem. Res. 19:1301–1310.
Edmond, J. (1992). Energy metabolism in developing brain cells. Can. J. Physiol. Pharmacol. 70:S118-S129.
Fitpatrick, S.M., Cooper, A.J.L., and Hertz, L. (1988). Effects of ammonia and β-methylene-DL-aspartate on the oxidation of glucose and pyruvate by neurons and astrocytes in primary culture. J. Neurochem. 51:1197–1203.
Garriga, J., and Russó, R. (1992). Effect of starvation on glycogen and glucose metabolism in different areas of the rat brain. Brain Res. 591:277–282.
Grossbard, L., and Schimke, R.T. (1966). Multiple hexokinases of rat tissues: purification and comparison of soluble forms. J. Biol. Chem. 241:3546–3560.
Hertz, L., and Peng, L. (1992). Energy metabolism at the cellular level of the CNS. Can. J. Physiol. Pharmacol. 70:S145-S157.
Hertz, L., Juurlink, B.H.J., Fosmark, H., and Schousboe, A. (1982). Methodological appendix: astrocytes in primary culture. In (S.E. Pfeiffer, ed.), Neuroscience Approached through Cell Culture, Vol. I, CRC, Boca Raton, FL, pp. 175–186.
Hertz, L., Juurlink, B.H.J., and Szuchet, S. (1985). Cell cultures. In (A. Lajtha, ed.), Handbook of Neurochemistry(2nd Ed.), Vol. 8, Plenum, New York, NY, pp. 603–661.
Hertz, L., Murthy, Ch.R.K., Lai, J.C.K., Fitzpatrick, S.M., and Cooper, A.J.L. (1987). Some metabolic effects of ammonia on astrocytes and neurons in primary cultures. Neurochem. Pathol. 6:97–129.
Hertz, L., Murthy, Ch.R.K., and Schousboe, A. (1988). Metabolism of glutamate and related amino acids. In (N.M. Norenberg, L. Hertz, and A. Schousboe, eds.), The Biochemical Pathology of Astrocytes, Alan R. Liss, New York, NY, pp. 395–406.
Kao-Jen, J., and Wilson, J.E. (1980). Localization of hexokinase in neural tissue: electron microscopic studies of rat cerebellar cortex. J. Neurochem. 35:667–678.
Lai, J.C.K. (1992). Oxidative metabolism in neuronal and non-neuronal mitochondria. Can. J. Physiol. Pharmacol. 70:S130-S137.
Lai, J.C.K., and Barrow, H.N. (1984). Comparison of the inhibitory effects of mercuric chloride on cytosolic and mitochondrial hexokinase activities in rat brain, kidney, and spleen. Comp. Biochem. Physiol. 78C:81–87.
Lai, J.C.K., and Behar, K.L. (1994). Differential inhibition of brain cytosolic & mitochondrial hexokinases by glucose-1,6-bisphosphate. FASEB J. 8: A820.
Lai, J.C.K., and Blass, J.P. (1984). Inhibition of brain glycolysis by aluminum. J. Neurochem. 42:438–446.
Lai, J.C.K., and Blass, J.P. (1985). Differences in the responses of brain cytosolic and mitochondrial hexokinases to three essential divalent metal ions. Comp. Biochem. Physiol. 80C:285–290.
Lai, J.C.K., and Clark, J.B. (1989). Isolation and characterization of synaptic and nonsynaptic mitochondria from mammalian brain. In (A.A. Boulton, G.K. Baker, and R.F. Butterworth, eds.), Neuromethods, Vol. 11, Humana Press, Clifton, NJ, pp. 43–98.
Lai, J.C.K., Baker, A., Carlson, K.C., Jr., and Blass, J.P. (1985). Differential effects of monovalent, divalent and trivalent metal ions on rat brain hexokinase. Comp. Biochem. Physiol. 80C:291–294.
Lai, J.C.K., Murthy, Ch.R.K., Cooper, A.J.L., Hertz, E., and Hertz, L. (1989). Differential effects of ammonia and β-methylene-DL-aspartate on metabolism of glutamate and related amino acids by astrocytes and neurons in primary culture. Neurochem. Res. 14:377–389.
Land, J.M., Booth, R.F.G., Berger, R., and Clark, J.B. (1977). Development of mitochondrial energy metabolism in rat brain. Biochem. J. 164:339–348.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., and Randall, R.J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–275.
Lusk, J.A., Manthorpe, C.M., Kao-Jen, J., and Wilson, J.E. (1980). Predominance of the cytoplasmic form of brain hexokinase in cultured astrocytes. J. Neurochem. 34:1412–1420.
Lynch, R.M., Fogarty, K.E., and Fay, F.S. (1991). Modulation of hexokinase association with mitochondria analyzed with quantitative three-dimensional confocal microscopy. J. Cell Biol. 112:385–395.
Rust, R.S., Jr., Carter J.G., Martin, D., Nerbonne, J.M., Lampe, P.A., Pusateri M.E., and Lowry, O.H. (1991). Enzyme levels in cultured astrocytes, oligodendrocytes and Schwann cells, and neurons from the cerebral cortex and superior cervical ganglia of the rat. Neurochem. Res. 16:991–999.
Wilkin, G. and Wilson, J.E. (1977). Localization of hexokinase in neural tissue: light microscopic studies with immunofluorescence and histochemical procedures. J. Neurochem. 29:1039–1051.
Wilson, J.E. (1980). Brain hexokinase: the prototype ambiquitous enzyme. In (B.L. Horecker, and E.R. Stadtman, eds.), Current Topics in Cellular Regulation, Vol. 16, Academic, New York, NY, pp. 1–54.
Wilson, J.E. (1983). Hexokinase. In (A. Lajtha, ed.), Handbook of Neurochemistry, Vol. 4 (2nd Ed.), Plenum, New York, NY, pp. 403–433.
Wilson, J.E. (1985). Regulation of mammalian hexokinase activity. In (R. Beitner, ed.), Regulation of Carbohydrate Metabolism, Vol. 1, CRC, Boca Raton, FL, pp. 45–85.
Yip, V., Carter, J.G., Dick, E., Rose, Z.B., and Lowry, O.H. (1985). Distribution of glucose-1,6-bisphosphate and IMP-activated glucose bisphosphatase in brain and retina. J. Neurochem. 44:1741–1746.
Yip, V., Pusateri, M.E., Carter, J., Rose, I.A., and Lowry, O.H. (1988). Distribution of glucose-1,6-bisphosphate system in brain and retina. J. Neurochem. 50:594–602.
Yu, A.C., Schousboe, A., and Hertz, L. (1982). Metabolic fate of 14C-labeled glutamate in astrocytes in primary culture. J. Neurochem. 39:954–960.
Yu, A.C.H., Hertz, E., and Hertz, L. (1984). Alterations in uptake and release for GABA, glutamate, and glutamine during biochemical maturation of highly purified cultures of cerebral cortical neurons of a GABAergic preparation. J. Neurochem. 42:951–960.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lai, J.C., Behar, K.L., Liang, B.B. et al. Hexokinase in Astrocytes: Kinetic and Regulatory Properties. Metab Brain Dis 14, 125–133 (1999). https://doi.org/10.1023/A:1020761831295
Issue Date:
DOI: https://doi.org/10.1023/A:1020761831295