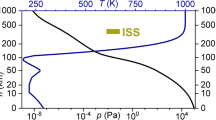
Abstract
During methane dissolution in water in a closed space, the pressure varies exponentially with time until the formation of methane hydrate. A model of this process is proposed that fits experimental data well. The methane concentration at the onset of hydrate formation is calculated as a function of temperature and pressure.
Similar content being viewed by others
REFERENCES
Martynets, V.G., Matizen, E.V., Kuskova, N.V., and Kukarin, V.F., Critical Line of (Xenon + Carbon Dioxide), J. Chem. Thermodyn., 1999, vol. 31, p. 191.
Bezverkhii, P.P., Kuskova, N.V., Martynets, V.G., and Matizen, E.V., Methane Hydrate Formation and Decomposition: Metastability Region and Phase-Equilibrium Curves, Khim. Interes. Ust. Razv., 1999, vol. 7, p. 643.
Maekava, T., Iton, S., Sakata, S., et al., Pressure and Temperature Conditions for Methane Hydrate Dissociation in Sodium Chloride Solutions, Geochem. J., 1995, vol. 29, p. 325.
Sychev, V.V., Vasserman, A.A., Zagoruchenko, V.A., et al., Termodinamicheskie svoistva metana (Thermodynamic Properties of Methane), Moscow: Izd. Standartov, 1979.
Istomin, V.A., Preduprezhdenie i likvidatsiya gazovykh gidratov v sistemakh sbora i promyslovoi obrabotki gaza i nefti (Prevention of Gas Hydration and Hydrate Liquidation in Industrial Gas and Oil Collection and Treatment Systems), Moscow: VNIIGaz, 1990.
Dil'man, V.V., Kulov, N.N., Lotkhov, V.A., et al., On the Difference in Rates of Absorption and Desorption of Gases, Teor. Osn. Khim. Tekhnol., 1998, vol. 32, no. 4, p. 377.
Landau, L.D. and Lifshits, E.M., Mekhanika sploshnykh sred (Continuum Mechanics), Moscow: Gostekhteorizdat, 1953.
Landau, L.D. and Lifshits, E.M., Statisticheskaya fizika (Statistical Physics), Moscow: Nauka, 1976.
Pau, P.C.F., Berg, J.O., and McMillan, W.G., Application of Stokes' Law to Ions in Aqueous Solution, J. Phys. Chem., 1990, vol. 94, no. 6, p. 2671.
Kaye, G.W. and Laby, T.H., Tables of Physical and Chemical Constants, London: Longmans, 1959, 12th ed. Translated under the title Tablitsy fizicheskikh i khimicheskikh postoyannykh, Moscow: Fizmatgiz, 1962.
Pic, J.-S., Herri, J.-M., and Cournil, M., Mechanisms of Methane Hydrate Crystallization in Semibatic Reactor, Ann. N. Y. Acad. Sci., 2000, vol. 912, p. 564.
Istomin, V.A. and Yakushev, V.S., Gazovye gidraty v prirodnykh usloviyakh (Gas Hydrates in Nature), Moscow: Nedra, 1992.
Handa, Y.P., Effect of Hydrostatic Pressure and Salinity on Gas Hydrates, J. Phys. Chem., 1990, vol. 94, no. 6, p. 2652.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martynets, V.G., Afanas'ev, I.S., Bezverkhii, P.P. et al. Equilibrium and Kinetic Properties of Methane Hydrate. Theoretical Foundations of Chemical Engineering 36, 495–499 (2002). https://doi.org/10.1023/A:1020686031117
Issue Date:
DOI: https://doi.org/10.1023/A:1020686031117